 |
||||||||||
Please read our latest articles at CLL Topics Updates!
Updates is an interactive site with an emphasis on current developments in the field.
Most Recent Article:
News you can use
(08/31/2012)
Important Notice About this Site
We suffered a transfiguring loss when Mr. P. C. Venkat, our Editor, Publisher and Chief Financial Officer, passed away from complications after an allogenic cord blood stem cell transplant in June of 2025.
It took a tremendous effort on his part to keep the non-profit organization running. Without his singular expertise and dedication, we knew we would be unable to fulfill the government regulatory requirements needed to maintain our non-profit status.
As of 2025, CLL Topics Inc., a non-profit 501(c) was dissolved and all its funds disbursed. We are now operating as CLL Topics LLC, a for-profit company incorporated in the state of Maryland. This, our former flagship site, is being retained on the Web as a resource to the public and to maintain historical continuity but is no longer actively updated.
We invite you to join us at our new, up-to-date site: CLL Topics Updates. It is an interactive site with an emphasis on current developments in the field.
We can think of no better testament to his life than to continue our mission in P.C.'s honor — and yours.
Recently Published
Older but Important
Many additional articles can be accessed from the
Table of Contents
 You can read the archives of our Topics Alert service.
You can read the archives of our Topics Alert service.
For the latest information on research, clinical trials and best practices — timely information that can make all the difference — please visit our new site: CLL Topics Updates.
Topics Alert was an email newsletter service offered from March 2025 through December 2025. An Alert went out whenever we had an interesting or important piece of information to communicate. If you would like to read through our archives, they can be seen here:
Link to the Alert Archive.

This site is dedicated to providing information and support to patients and families dealing with chronic lymphocytic leukemia (CLL). It takes a clearly patient-oriented view of the world. The subject matter is focused on current developments in understanding and treating the disease.
Discussion topics include basic science, therapeutic approaches, clinical trials and analyses of the results of these trials. We are witnessing significant changes in our understanding of the processes involved and in the therapies available to combat the disease. It is hoped that the discussions in our articles will bridge the gap between the professional cancer research community and practicing oncologists on the one hand and patients and their families on the other.
The opinions and analyses presented are those of lay observers of the CLL scene and not those of medical or research professionals. They are not intended to provide clinical guidance or recommendations on the management of the disease. This site draws its strength from a team of dedicated volunteers. None of us has any affiliation with the medical or pharmaceutical industries. Our only interest is patient education and advocacy. We hope our website can help you understand your therapy options better and make you better prepared to discuss these options with your healthcare providers.
You may use the Contents page to review the material in this site organized by major subject area or use the customized site search form in the right column to get quickly to a specific topic of interest.
We welcome your comments about this website or your thoughts about any of the subjects discussed or referenced here. Please use the form on our Feedback page or send your email to us at ![]() .
.
Chaya & P. C. Venkat
Founders

CLL Topics.org was the online publication of a patient-operated, non-profit corporation, CLL Topics, Inc.
We have no affiliation with any entity in the health care industry and do not accept any advertising on our publication. Our operating expenses were funded entirely by CLL Topics, Inc. which derives all its revenues from donations from the public. The bulk of funds that were raised from our donors was devoted to supporting clinical trials for therapies in CLL. For details, please use links to financial statements and other corporate documents below.
Please refer to the About Us section on this website for background information on the CLL Topics Team.
Please do remember that the writers of the articles on CLLTopics.org are not medical professionals and do not provide medical advice or recommendations for treatment. Please consult and follow the advice of your own healthcare providers.
Co-founders:
Chief Science Writer:
Chaya Venkat
Editor, Publisher and Webmaster:
P. C. Venkat
Board of Directors:
L. Keith Friedlander
Wendy Ramsey
Chaya Venkat
P. C. Venkat
Each member of our Board is a CLL patient or family member of a CLL patient. For short biographies of our Board members, please see the About Us page on this website.

According to Mayo
You can read an article about CLL Topics and its founders in the Spring 05 issue of Mayo Magazine: Information Empowers Leukemia Patients
According to
the Wall Street Journal
The Journal, too, has had occasion to publish an article in which CLL Topics figured. You can read the March 2025 article by Journal reporter Rhonda Rundle at: Testing the Power of Green Tea.
According to
the Washington Post
The Washington Post was not far behind in publishing an article in which CLL Topics received positive coverage. You can read read why their reporter says, "For the latest real news specific to the condition, I would find the best stuff on CLL Topics." Here is a link to the Post article, A 10-Year Checkup. If you find this link does not work, write to us and we will help you locate one that does.

Our Non-Discrimination Policy
We Support CLL Patients Everywhere - and Bar None
CLL Topics extends its services to and encourages participation in its activities by all individuals affected by Chronic Lymphocytic Leukemia. It does not discriminate on the basis of race, ethnicity, religion, age, size, national origin, citizenship, geographic location, language, gender, sexual orientation, socioeconomic background or mental or physical handicap. In fact, we are pleased to extend our services to any person who wishes to benefit from them.

An Open Invitation
Exercise Your Pen - or Keyboard
Your Opinion Counts
The CLL Topics Team is happy to hear from you. We value your willingness to share your knowledge, experience and concern for our community. The section titled "Patients' Corner" is intended to provide a forum for your opinions and insights. Of course, your articles can be published in any of the other sections if they fit better there. If you would like us to consider for publication an article you have written, please submit the document by email to the address shown below.
Don't forget to include your name and email address or phone number so that we can contact you if we have any questions. We will entertain full-length articles, announcements or snippets of information or commentary that may be of interest to our readers.
— Editor
![]()
Volunteer Patient Support Groups
You will find links to a number of patient education and support groups on our page dedicated to Support Groups.
Navigating Topics
Help to Get the Most Out of This Site
It's Easier Than You Think
The sheer amount of information available on this site may be overwhelming at first. But appearances can be deceptive, especially in this case. Most people find that there are some easy ways of getting around the site, locating topics of interest, printing the material they want as hard copies, downloading material and solving technical problems as they arise. We have paid special attention to browser compatibility, accessibility and ease of use. Many of these subjects are covered in our Navigation Notes article.
Of course, we can always use your help to identify those areas which have still escaped our attention, or simply those that do not work right for you. Just write to us with a clear explanation of the problem and include the basic details of your computer set-up. It is entirely possible that someone else has run into your particular problem and we may already have an off-the-shelf solution. We do not promise to cure all the technical ills of the world wide web. We will, however, listen to your problems politely and at least try to make helpful suggestions. After all, we would like to make our work accessible to as many people as possible. That's why we started Topics in the first place.

The Contents page will provide you with links to all the main subject areas on this website dealing with the disease, its diagnosis, prognosis, therapies, clinical research and the societal aspects of coping with the disease and the medical establishment.
A CLL Primer is directed at newly diagnosed patients (or as a refresher for those desiring to get caught up quickly) and serves to orient the patient or caregiver and highlight some basic facts and considerations. A list of additional articles to read is suggested in Step 5.
You will find spreadsheet templates you can use to keep track of your clinical data in Your Charts.
Our Reference section has links to many important resources on the Web for patients seeking to expand their knowledge of the disease.
For those with a scholastic bent, the Textbooks section provides links to a collection of superb online textbooks that can provide an entire education.
The Search Tools page gives you access to professional search tools to locate abstracts and articles, search scientific databases, medical news and clinical trials.
Donations: Your generous support and encouragement have been terrific. You have indeed shown us that you think what we do is worthwhile. With your help we have exceeded our fundraising goals for the year. As of July 1, 2025 we are temporarily suspending our fundraising. This also happens to be the time when our editor, publisher, webmaster and record keeper is going into a chemoimmunotherapy treatment program - and suspending fundraising will enable us to concentrate our resources on running our publishing operation (this website and Topics Alert). Send us an email (![]() ) if you would like to volunteer your time and skills to join us in the work of this organization.
) if you would like to volunteer your time and skills to join us in the work of this organization.
Please do browse Current Articles and Contents to orient yourself to the resources available on this site. The material presented is based on serious, peer-reviewed articles in professionals and scientific journals, complete with references and links to source materials. However, we have made it a priority to simplify the language and clarify the communication to make it accessible to the lay reader. Most important, we offer critical review form the patient's point of view. We hope you will become familiar with the terminology, our style of presentation and the navigation of this website pretty quickly. If you do experience difficulty with any part of it, please send us your comments so that we may improve this website. You may use the form on our Feedback page or send email to ![]() .
.

Company News
CLL Topics becomes a Corporation.
A Short History
This website, CLL Topics.org, has been on the Web for a relatively short time. It was launched on April 4, 2025.
CLL Topics, Inc. was registered as an Arizona non-profit corporation on October 27, 2025. The IRS issued an advance ruling on the Corporation's 501(c)(3) tax-exempt status on June 21, 2025.
On December 16, 2025 the collaboration with Mayo Clinic on Project Alpha was announced. As of December 16, 2025, the Corporation started fundraising to support clinical trials. For more details please see our Donations page.
On February 25, 2025, the IRS confirmed its advance ruling that CLL Topics, Inc. is a public charity, having conformed to all regulatory requirements for this status. As of March 1, 2025, the Corporation has finished five years of regulatory reporting, raised over $300,000 in funds from worldwide donations principally for supporting clinical research in CLL, sponsored and funded four clinical trials with a fifth in the final stages of regulatory approval and maintained an active online publication and bulletin system in the form of CLLTopics.org and Topics Alert. On February 25, 2025, we launched an online journal focusing on the details of a cord blood stem cell transplant for a CLL patient.

As of December 31, 2025 we have received a total of $310,828.34 from 555 donors. For details and a list of donors, please see our Acknowledgements page.
We have fully funded our commitments to three clinical trials that we have sponsored, and another two are pending disbursement. Current fund balances will support investigations of a number of promising new agents. Additional details will be reported as we develop these projects with the research community. You can read the details of past projects we have sponsored on the page on Sponsored Projects. Your donations have enabled us to offer critical financial support to these investigations at a formative stage and demonstrate patient involvement and reflect patient priorities in the process.
We have reached our fundraising goals for the year 2025 and as of July 1, 200 we will temporarily suspend fundraising activities while our publisher, webmaster and chief administrator is undergoing treatment for his CLL. Suspending this labor-intensive activity will lighten up our operational commitments and allow us to concentrate our time and effort on maintaining our publishing and educational activities, including this website and Topics Alerts. We will report on the results from our sponsored projects as and when they become available.
We are grateful to all our generous donors for their support and encouragement.


Financial Statements csgo betting
To see financial statements for the Corporation you may use the following links:
Other Corporate Documents
Additional corporate information may be found through the following links:
Annual Report to the Board for 2025;
Annual Report to the Board for 2025;
Annual Report to the Board for 2025;
Annual Report to the Board for 2025;
Ruling Confirming Status as Public Charity;
Application for Tax Exemption;
Arizona Secretary of State Filings;
Arizona Corporation Commission Filings.
Our Site Is Standards Compliant
 |
 |


|
Both search forms shown here will return results from customized searches of this website. The Google search form above produces results unencumbered by sponsored ads, while the search results from the Atomz engine below come with sponsored ads which some will find annoying. However, it has the advantage of more frequent indexing and a more up-to-date database — so you are more likely to find material recently added to the website.

There is also an advanced search form for more complex searches. You are encouraged to experiment with these search tools.
All materials contained on this site are protected by United States copyright law and may not be reproduced, distributed, transmitted, displayed, published or broadcast without the prior written permission of CLL Topics, Inc. You may not alter or remove any trademark, copyright or other notice from copies of the content.
However, you may download and print material from CLLTopics.org exclusively for your personal, noncommercial use.
Copyright © 2025 to 2025.
CLL Topics, Inc.
All Rights Reserved.

P. C. Venkat, 1949-2008
We are deeply saddened to announce the death of our Webmaster, Editor and Publisher.
P. C. Venkat passed away on June 23rd from complications after an allogenic cord blood stem cell transplant. You can read the details of his transplant journey at Harvey's Journal.
After a period of mourning, CLL Topics Updates has continued our mission in his honor. (7/3/2008)
Veltuzumab
Fully Humanized Competition for Rituxan
Another Anti-CD20 Monoclonal in Clinical Trials
 Immunomedics is recruiting CLL and NHL patients for a Phase I/II clinical trial of their new anti-CD20 monoclonal at multiple centers across the U.S. Read about how this Mab compares to the gold standard Rituxan in Veltuzumab: Another Anti-CD20 Monoclonal in Clinical Trials. (9/21/2008)
Immunomedics is recruiting CLL and NHL patients for a Phase I/II clinical trial of their new anti-CD20 monoclonal at multiple centers across the U.S. Read about how this Mab compares to the gold standard Rituxan in Veltuzumab: Another Anti-CD20 Monoclonal in Clinical Trials. (9/21/2008)
NK-cell Therapy: Better than a transplant?
GVL without GVHD?
Having Your Cake and Eating It Too
 New research findings point to NK (Natural Killer) cells as the most potent cell line capable of GVL (graft-versus-leukemia), but these cells do not seem to do as much GVHD (Graft-Versus-Host-Disease). A brand new clinical trial launched at the University of Minnesota uses haplo NK cells in an attempt to get the much desired GVL, while steering clear of the dreaded GVHD. Our review NK-cell Therapy: Better than a transplant? describes the background, rationale and advantages of this approach. (7/28/2008)
New research findings point to NK (Natural Killer) cells as the most potent cell line capable of GVL (graft-versus-leukemia), but these cells do not seem to do as much GVHD (Graft-Versus-Host-Disease). A brand new clinical trial launched at the University of Minnesota uses haplo NK cells in an attempt to get the much desired GVL, while steering clear of the dreaded GVHD. Our review NK-cell Therapy: Better than a transplant? describes the background, rationale and advantages of this approach. (7/28/2008)
Long Term Survival
Are CLL Patients Living Longer?
If Not a Cure, At Least Longer Life?
 A new article has been published in Blood Journal on the subject of long term survival
of CLL patients. The paper, based on SEER survey data, is reviewed in our article titled
Are CLL Patients Living Longer? (3/2/2008)
A new article has been published in Blood Journal on the subject of long term survival
of CLL patients. The paper, based on SEER survey data, is reviewed in our article titled
Are CLL Patients Living Longer? (3/2/2008)
Single Agent Fludarabine: Definitely Not the Gold Standard
UK CLL4 Clinical Trial Results
AIHA: One More Reason to Shun Single Agent Fludarabine
 Fludarabine and chlorambucil as single agents have often been the conventional
choice for first line treatment of CLL even while there was ample evidence that combination
therapies produced more impressive responses. Now we have results from the UK CLL4
clinical trial, a head-to-head comparison of these same single agents against a combination of
fludarabine and cyclophosphamide. In
Single Agent Fludarabine: Definitely Not the Gold Standard, we examine the role of fludarabine
monotherapy in precipitating potentially life threatening AIHA. (2/09/08)
Fludarabine and chlorambucil as single agents have often been the conventional
choice for first line treatment of CLL even while there was ample evidence that combination
therapies produced more impressive responses. Now we have results from the UK CLL4
clinical trial, a head-to-head comparison of these same single agents against a combination of
fludarabine and cyclophosphamide. In
Single Agent Fludarabine: Definitely Not the Gold Standard, we examine the role of fludarabine
monotherapy in precipitating potentially life threatening AIHA. (2/09/08)
MUD Transplants at the NCI
NIH Will Pick Up the Tab
Brand New Protocol Makes Its Debut
 The NCI has just announced a new mini-allo MUD stem cell transplant clinical trial
designed to evaluate the relative efficacy of two different methods of handling GVHD
(graft versus host disease). Patients should take additional – and particular
– notice of the fact that the NCI will pay for the entire protocol – from
cytoreduction to transplant and post-transplant monitoring. Patients with a well
matched unrelated donor (MUD) but without adequate insurance will find this trial
may open a door that is otherwise closed. To find out more please read
MUD Transplants at the NCI. (1/18/08)
The NCI has just announced a new mini-allo MUD stem cell transplant clinical trial
designed to evaluate the relative efficacy of two different methods of handling GVHD
(graft versus host disease). Patients should take additional – and particular
– notice of the fact that the NCI will pay for the entire protocol – from
cytoreduction to transplant and post-transplant monitoring. Patients with a well
matched unrelated donor (MUD) but without adequate insurance will find this trial
may open a door that is otherwise closed. To find out more please read
MUD Transplants at the NCI. (1/18/08)
Treanda: A Facelift for Bendamustine
Old Drug Recycled as New Therapy
Cephalon Makes a Splash with Orphan Drug for CLL
 Recent presentations at the ASH 2025 meeting and a flurry of press releases on
Treanda (bendamustine hydrochloride)
have raised a considerable level of interest in the drug. While the agent itself has been
around for over thirty years, its development as a drug for use in CLL is more recent.
In our article, Treanda: A Facelift for Bendamustine,
we examine the results of clinical trials reported to date and offer our analysis of the
clinical trial results and the potential value of this drug in treating CLL. (12/15/07)
Recent presentations at the ASH 2025 meeting and a flurry of press releases on
Treanda (bendamustine hydrochloride)
have raised a considerable level of interest in the drug. While the agent itself has been
around for over thirty years, its development as a drug for use in CLL is more recent.
In our article, Treanda: A Facelift for Bendamustine,
we examine the results of clinical trials reported to date and offer our analysis of the
clinical trial results and the potential value of this drug in treating CLL. (12/15/07)
ITP: Immune Thrombocytopenia
Possible Connection to H. pylori
Platelets in Danger
 Immune thrombocytopenia (ITP) is a serious complication of CLL that affects a small
percentage of patients. It is a condition in which the immune system destroys platelets.
Research suggests that a common pathogen, Helicobacter pylori, can sometimes be the cause of ITP
and that controlling and eradicating this infection can resolve ITP in its early stage.
We discuss this and other characteristics of this condition in
ITP: Immune Thrombocytopenia. (12/6/07)
Immune thrombocytopenia (ITP) is a serious complication of CLL that affects a small
percentage of patients. It is a condition in which the immune system destroys platelets.
Research suggests that a common pathogen, Helicobacter pylori, can sometimes be the cause of ITP
and that controlling and eradicating this infection can resolve ITP in its early stage.
We discuss this and other characteristics of this condition in
ITP: Immune Thrombocytopenia. (12/6/07)
NCI Overview on CLL
Up-to-date and Comprehensive Statement
Official — and Exceptionally Useful
 The National Cancer Institute produces a variety of publications that are useful – and generally taken for granted. We would like to alert you to a newly updated document that is on the NCI website: Chronic Lymphocytic Leukemia (PDQ®): Treatment. This is an up-to-date, comprehensive, authoritative reference with an extensive bibliography that should prove useful to professionals and patients. (There is a simplistic patient version and a more businesslike health-professional version.) Highly recommended reading. (10/21/07)
The National Cancer Institute produces a variety of publications that are useful – and generally taken for granted. We would like to alert you to a newly updated document that is on the NCI website: Chronic Lymphocytic Leukemia (PDQ®): Treatment. This is an up-to-date, comprehensive, authoritative reference with an extensive bibliography that should prove useful to professionals and patients. (There is a simplistic patient version and a more businesslike health-professional version.) Highly recommended reading. (10/21/07)
Refractory CLL
When You Have Been Around the Track a Few Times
Salavge Therapy Produces Grim Statistics
 There are not many good therapy choices for CLL patients who become resistant
to fludarabine and Campath. While a number of salvage therapies
are in use, the results are variable and the survival statistics are grim. We
agree with the experts in the field that a wider range of approaches and more
effective agents are desperately needed for these patients.
Refractory CLL is our review the literature
and the latest reported results of salvage treatment for refractory CLL. (10/14/07)
There are not many good therapy choices for CLL patients who become resistant
to fludarabine and Campath. While a number of salvage therapies
are in use, the results are variable and the survival statistics are grim. We
agree with the experts in the field that a wider range of approaches and more
effective agents are desperately needed for these patients.
Refractory CLL is our review the literature
and the latest reported results of salvage treatment for refractory CLL. (10/14/07)
Improving Flu Vaccinations
Launch of Phase II Clinical Trial Sponsored by CLL Topics
Jab & Dab: At Last We Have Liftoff!
 The clinical trial we first announced in our May 13, 2025 article,
Improving Routine Immunizations,
has finally cleared all hurdles and the research team at the Royal Bournemouth
Hospital is ready to recruit patients for this controlled Phase II clinical trial.
The trial is designed to compare the results from imiquimod-enhanced flu
shots to results from an age-matched control group. We discuss the
science, trial protocol and enrollment criteria in a new article titled
Jab & Dab: We Have Liftoff!.
The patient community has tremendous “ownership” in this trial: the concept
originated at CLL Topics, and it is sponsored with your donation money! (10/6/07)
The clinical trial we first announced in our May 13, 2025 article,
Improving Routine Immunizations,
has finally cleared all hurdles and the research team at the Royal Bournemouth
Hospital is ready to recruit patients for this controlled Phase II clinical trial.
The trial is designed to compare the results from imiquimod-enhanced flu
shots to results from an age-matched control group. We discuss the
science, trial protocol and enrollment criteria in a new article titled
Jab & Dab: We Have Liftoff!.
The patient community has tremendous “ownership” in this trial: the concept
originated at CLL Topics, and it is sponsored with your donation money! (10/6/07)
Quality of Life with CLL
First of Several Articles Published online casino nz
Results from Mayo Clinic Survey
 We would like to thank many of our readers who heeded our call and participated in the Quality of Life survey
conducted by the CLL research team at Mayo Clinic, Rochester, MN. The results from this
survey continue to be analyzed but some of the early findings have been published
in an article which appeared in the British Journal of Haematology in September 2025.
For more details, refer to
Topics Alert #253: Quality of Life with CLL. (9/29/07)
We would like to thank many of our readers who heeded our call and participated in the Quality of Life survey
conducted by the CLL research team at Mayo Clinic, Rochester, MN. The results from this
survey continue to be analyzed but some of the early findings have been published
in an article which appeared in the British Journal of Haematology in September 2025.
For more details, refer to
Topics Alert #253: Quality of Life with CLL. (9/29/07)
Campath as Single-Agent Frontline Therapy
FDA Expands Approval
Jumping the Gun
 The FDA approved Campath for use in treating relapsed or refractory CLL in May 2025.
The drug has since proved useful, especially in those cases with p53 deletions which are otherwise
resistant to treatment. However, the antibody produces severe immune suppression and other
side effects and its use has been limited. Most recently, based on the results of a
clinical trial that compared Campath with chlorambucil, the FDA has expanded the use of
the drug to include frontline single-agent treatment of CLL. In this article,
Jumping the Gun, we examine the value of the broader
use of Campath as frontline single-agent therapy for CLL. (9/24/07)
The FDA approved Campath for use in treating relapsed or refractory CLL in May 2025.
The drug has since proved useful, especially in those cases with p53 deletions which are otherwise
resistant to treatment. However, the antibody produces severe immune suppression and other
side effects and its use has been limited. Most recently, based on the results of a
clinical trial that compared Campath with chlorambucil, the FDA has expanded the use of
the drug to include frontline single-agent treatment of CLL. In this article,
Jumping the Gun, we examine the value of the broader
use of Campath as frontline single-agent therapy for CLL. (9/24/07)
Opinion: What Say You, Dr. Expert?
Viewpoints from Noted CLL Experts
Making Sensible Decisions About Stem Cell Transplants
 The clinical research community is slowly coming to grips with the question of when a stem cell
transplant makes sense in CLL. The mortality and GVHD risks associated with transplants are
definite deterrents but there are substantial survival risks, too, in not taking
definitive action when faced with aggressive, poor-prognostic or refractory disease.
Four of our favorite CLL experts from major research centers have been generous in sharing
their personal viewpoints on the question. You can read their opinions in our article,
"What Say You, Dr. Expert?" (9/10/07)
The clinical research community is slowly coming to grips with the question of when a stem cell
transplant makes sense in CLL. The mortality and GVHD risks associated with transplants are
definite deterrents but there are substantial survival risks, too, in not taking
definitive action when faced with aggressive, poor-prognostic or refractory disease.
Four of our favorite CLL experts from major research centers have been generous in sharing
their personal viewpoints on the question. You can read their opinions in our article,
"What Say You, Dr. Expert?" (9/10/07)
Radioimmunotherapy: Underappreciated Option?
Hot and Cold Monoclonal Antibodies
A Protocol Waiting for a Trial
 There are two approved radioimmunotherapy drugs currently on the market, Bexxar and Zevalin.
Unfortunately, both are at risk of becoming unavailable to patients as a result of reimbursement
issues. Both drugs have proven effective at producing durable remissions in follicular lymphoma
and one, Bexxar, is currently being tested in CLL at the Fred Hutchinson Cancer Research Center.
In our article Radioimmunotherapy: Underappreciated
Option?, we suggest an approach that may be used in early stage patients to reduce bone
marrow involvement and then deliver clean, durable remissions: all in a patient-friendly protocol.
We can use your help in getting the research community interested in launching a clinical trial
to test the concept.(9/6/07)
There are two approved radioimmunotherapy drugs currently on the market, Bexxar and Zevalin.
Unfortunately, both are at risk of becoming unavailable to patients as a result of reimbursement
issues. Both drugs have proven effective at producing durable remissions in follicular lymphoma
and one, Bexxar, is currently being tested in CLL at the Fred Hutchinson Cancer Research Center.
In our article Radioimmunotherapy: Underappreciated
Option?, we suggest an approach that may be used in early stage patients to reduce bone
marrow involvement and then deliver clean, durable remissions: all in a patient-friendly protocol.
We can use your help in getting the research community interested in launching a clinical trial
to test the concept.(9/6/07)
Radioimmunotherapy Reimbursement Cuts
Patients Appeal Against Proposed Changes
An Important and Effective Therapy Option May Disappear
 We would like to draw your attention to an issue that threatens the availability of a valuable,
FDA approved therapy for B-cell malignancies. Proposed modifications to the
Medicare/Medicaid reimbursement schedules would make it uneconomic for hospitals
to continue offering radioimmunotherapy as an option for treating diseases like
the lymphomas and CLL. This comes at a time when a promising new clinical trial
is under way at the Hutch to establish a methodology for using radioimmunotherapy
agents effectively in CLL. See Radioimmunotherapy
Reimbursement Cuts, our announcement on this issue and please take the time to
add your endorsement to the patient initiative that has been launched to keep this therapy option alive. (8/31/07)
We would like to draw your attention to an issue that threatens the availability of a valuable,
FDA approved therapy for B-cell malignancies. Proposed modifications to the
Medicare/Medicaid reimbursement schedules would make it uneconomic for hospitals
to continue offering radioimmunotherapy as an option for treating diseases like
the lymphomas and CLL. This comes at a time when a promising new clinical trial
is under way at the Hutch to establish a methodology for using radioimmunotherapy
agents effectively in CLL. See Radioimmunotherapy
Reimbursement Cuts, our announcement on this issue and please take the time to
add your endorsement to the patient initiative that has been launched to keep this therapy option alive. (8/31/07)
FISHing in Murky Waters
The Story of Undetected FISH Defects
The Importance of Asking the Meaningful Question
 The typical commercially available FISH panel contains four probes which seek to establish
the presence or absence of the most common cytogenetic abnormalities in CLL. Results that
do not have one of these four common deletions or mutations are labeled “normal” —
a misleading label. CLL cells almost certainly suffer from some genetic abnormality that
makes them cancer cells to begin with. Even for those patients who have one or more of the
known defects, the limited probe set could mask additional defects that may have a bearing
on risk assessment and treatment. In FISHing in Murky Waters,
we examine the importance of expanding the probe set presently used in the standard CLL FISH panel. (8/29/07)
The typical commercially available FISH panel contains four probes which seek to establish
the presence or absence of the most common cytogenetic abnormalities in CLL. Results that
do not have one of these four common deletions or mutations are labeled “normal” —
a misleading label. CLL cells almost certainly suffer from some genetic abnormality that
makes them cancer cells to begin with. Even for those patients who have one or more of the
known defects, the limited probe set could mask additional defects that may have a bearing
on risk assessment and treatment. In FISHing in Murky Waters,
we examine the importance of expanding the probe set presently used in the standard CLL FISH panel. (8/29/07)
Cord Blood Transplants
Review of the State of the Art
Greatly Improved Odds: but Transplant Mortality Remains
 The state of the art in cord blood transplants has come a long way in the past few years. Originally attempted only with children because of the limited stem cell dose available in umbilical cords, these procedures have evolved to include double cords and are now producing results in adults comparable with those from adult donor transplants. We review the results from the University of Minnesota and elsewhere and explain the concepts involved in our article titled Cord Blood Transplants: State of the Art. For many patients who do not have a matched sibling or unrelated adult donor, cord blood transplants are a lifeline of hope for a durable remission and perhaps a cure. (8/11/07)
The state of the art in cord blood transplants has come a long way in the past few years. Originally attempted only with children because of the limited stem cell dose available in umbilical cords, these procedures have evolved to include double cords and are now producing results in adults comparable with those from adult donor transplants. We review the results from the University of Minnesota and elsewhere and explain the concepts involved in our article titled Cord Blood Transplants: State of the Art. For many patients who do not have a matched sibling or unrelated adult donor, cord blood transplants are a lifeline of hope for a durable remission and perhaps a cure. (8/11/07)
Catch 22 – The Transplant Minefield
The Chance of a Real Cure versus Transplant Mortality Risk
Expert Opinions Diverge on Timing
 Many CLL patients with adverse prognostic indicators know that sooner or later they will have to face
the prospect of a stem cell transplant. Current research shows improving results from mini-allo transplants
for CLL patients, especially if they have clean remissions and are otherwise in good health going into the
transplants. While this might argue for early, elective transplants for some patients, the mortality risks
are still significant and there is as yet no consensus among transplant experts on timing and exhaustion
of other therapy options. To understand the issues involved, please read our case study of an anonymous
patient volunteer in Catch 22 – The Transplant Minefield. (7/4/07)
Many CLL patients with adverse prognostic indicators know that sooner or later they will have to face
the prospect of a stem cell transplant. Current research shows improving results from mini-allo transplants
for CLL patients, especially if they have clean remissions and are otherwise in good health going into the
transplants. While this might argue for early, elective transplants for some patients, the mortality risks
are still significant and there is as yet no consensus among transplant experts on timing and exhaustion
of other therapy options. To understand the issues involved, please read our case study of an anonymous
patient volunteer in Catch 22 – The Transplant Minefield. (7/4/07)
Strategy & Tactics
Harvey Chooses to Fight on His Own Terms
Battle Plans
 Harvey, our purely hypothetical patient with challenging cytogenetics, is not unique in facing
a complex and aggressive disease. However, he is determined to be as organized and logical about
it as he can and use his resources to the best effect. He has put together a plan that may give
him a chance to beat the disease and live to be an old man. Read about his battle plans in
Strategy and Tactics. (6/12/07)
Harvey, our purely hypothetical patient with challenging cytogenetics, is not unique in facing
a complex and aggressive disease. However, he is determined to be as organized and logical about
it as he can and use his resources to the best effect. He has put together a plan that may give
him a chance to beat the disease and live to be an old man. Read about his battle plans in
Strategy and Tactics. (6/12/07)
Incidentally, we would also like to tell you about future changes in our operations and fundraising efforts here at CLL Topics.
Radioimmunotherapy
The Tricky Business of Using Radiation in CLL
Bexxar: An Introduction
 A Phase II clinical trial for CLL has been launched by the Fred Hutchinson Cancer Research Center in
Seattle, using the radioimmunotherapy agent, Bexxar. By combining a CD20-targeting monoclonal antibody
with a radioactive payload, this agent offers the promise of high-precision targeting plus
a lethal wallop – which may lead to low MRD and long remissions. But since CLL invariably involves
the bone marrow, the partcipants must first go through a course of RF therapy to get B-cell infiltration
of the marrow as low as possible and thereby avoid damage to the bone marrow and precious stem cells.
Read about how this first-line therapy may work for you in
Bexxar: An Introduction to Radioimmunotherapy. (5/15/07)
A Phase II clinical trial for CLL has been launched by the Fred Hutchinson Cancer Research Center in
Seattle, using the radioimmunotherapy agent, Bexxar. By combining a CD20-targeting monoclonal antibody
with a radioactive payload, this agent offers the promise of high-precision targeting plus
a lethal wallop – which may lead to low MRD and long remissions. But since CLL invariably involves
the bone marrow, the partcipants must first go through a course of RF therapy to get B-cell infiltration
of the marrow as low as possible and thereby avoid damage to the bone marrow and precious stem cells.
Read about how this first-line therapy may work for you in
Bexxar: An Introduction to Radioimmunotherapy. (5/15/07)
GVHD Developments
A New Agent to Improve Chances of Survival
GVHD: A Pebble in the Transplant Shoe
 While mini allo stem cell transplants hold the promise of a cure for this disease, the risk of
mortality and graft versus host disease have been major deterrents to using this technique in high risk
CLL patients. New results from research in managing the worst effects of GVHD point the way to improved
survival and transplant related symptoms. We review encouraging results reported by the team at the
Fred Hutchinson Cancer Research Center in our article
GVHD: A Pebble in the Transplant Shoe
and explain why this could be very important to CLL patients with poor prognostics. (5/3/07)
While mini allo stem cell transplants hold the promise of a cure for this disease, the risk of
mortality and graft versus host disease have been major deterrents to using this technique in high risk
CLL patients. New results from research in managing the worst effects of GVHD point the way to improved
survival and transplant related symptoms. We review encouraging results reported by the team at the
Fred Hutchinson Cancer Research Center in our article
GVHD: A Pebble in the Transplant Shoe
and explain why this could be very important to CLL patients with poor prognostics. (5/3/07)
Therapy Choices
How Much Time Do You Think You Have?
Jenny Tells the Forest Story
 Forest Bump is the pen name of a CLL patient who volunteered to share with our readers his experience in the
FCR Lite clinical trial. You can read the first
installment of this case study in
Forest Bump. As it sometimes happens with CLL, what followed next was not something he or his
wife, Jenny, or indeed any of his doctors, anticipated. Forest suffered complications brought on by his CLL
and died in early March. Jenny bravely picks up the tale here, in the conclusion of the
Forest Bump saga titled
Jenny Tells the Forest Story. Easy answers are hard to come by but the story may provide you with some
serious food for thought. (4/23/07)
Forest Bump is the pen name of a CLL patient who volunteered to share with our readers his experience in the
FCR Lite clinical trial. You can read the first
installment of this case study in
Forest Bump. As it sometimes happens with CLL, what followed next was not something he or his
wife, Jenny, or indeed any of his doctors, anticipated. Forest suffered complications brought on by his CLL
and died in early March. Jenny bravely picks up the tale here, in the conclusion of the
Forest Bump saga titled
Jenny Tells the Forest Story. Easy answers are hard to come by but the story may provide you with some
serious food for thought. (4/23/07)
Immunotoxins
Monoclonal Antibodies with Enhanced Firepower
CAT8015 – A Trojan Horse
 Investigators at the NCI and NIH are hard at work on another agent that promises to combine sharp targeting
and a powerful knockout punch to destroy CLL cells: an immunotoxin called CAT-8015. A new clinical trial
with this agent has been announced and is currently recruiting. In our article,
CAT8015 – A Trojan Horse, we review the
background science and discuss the pros and cons of this approach. (4/12/07)
Investigators at the NCI and NIH are hard at work on another agent that promises to combine sharp targeting
and a powerful knockout punch to destroy CLL cells: an immunotoxin called CAT-8015. A new clinical trial
with this agent has been announced and is currently recruiting. In our article,
CAT8015 – A Trojan Horse, we review the
background science and discuss the pros and cons of this approach. (4/12/07)
Antibody in Development
An Antibody Targeted to the CD23 Marker
Lumiliximab: The Next Drug in CLL?
 Biogen Idec, the folks that brought us Rituxan, are busy developing a new monoclonal antibody for use in CLL.
The drug, Lumiliximab (formerly IDEC 152), has some interesting features that might make it useful in treating CLL.
It targets CD23, a marker which is brightly expressed by CLL cells and is in fact a B-cell activation marker.
The drug appears to have an enviably low toxicity profile. We review the research literature from early clinical
trials, discuss the major features of the agent itself and evaluate a recently launched pivotal Phase III
multicenter clinical trial of the drug as an addition to the FCR protocol. For the details, see our review
titled Lumiliximab: The Next Drug in CLL? (3/6/07)
Biogen Idec, the folks that brought us Rituxan, are busy developing a new monoclonal antibody for use in CLL.
The drug, Lumiliximab (formerly IDEC 152), has some interesting features that might make it useful in treating CLL.
It targets CD23, a marker which is brightly expressed by CLL cells and is in fact a B-cell activation marker.
The drug appears to have an enviably low toxicity profile. We review the research literature from early clinical
trials, discuss the major features of the agent itself and evaluate a recently launched pivotal Phase III
multicenter clinical trial of the drug as an addition to the FCR protocol. For the details, see our review
titled Lumiliximab: The Next Drug in CLL? (3/6/07)
Refractory CLL
Celgene Targets Revlimid as CLL Therapy
Revlimid to the Rescue?
 Oncology drug maker, Celgene Corp., sponsored two recent phase II clinical trials
for the use of its drug Revlimid, in CLL. The results of these trials were reported
in the 2025 ASH annual meeting in Orlando. In our article,
Revlimid to the Rescue?,
we review the results of these trials and present our commentary on the drug's efficacy,
adverse effects and potential value in cases with difficult prognostic factors. Any CLL
patient who is asked to consider off label use of this drug or to participate in
Celgene's big follow-up phase III clinical trial needs to do his or her homework first
— starting by reading this article. (1/30/07)
Oncology drug maker, Celgene Corp., sponsored two recent phase II clinical trials
for the use of its drug Revlimid, in CLL. The results of these trials were reported
in the 2025 ASH annual meeting in Orlando. In our article,
Revlimid to the Rescue?,
we review the results of these trials and present our commentary on the drug's efficacy,
adverse effects and potential value in cases with difficult prognostic factors. Any CLL
patient who is asked to consider off label use of this drug or to participate in
Celgene's big follow-up phase III clinical trial needs to do his or her homework first
— starting by reading this article. (1/30/07)
HuMax-CD20 for Follicular Lymphoma
Phase III Clinical Trial of Single-Agent HuMax-CD20
Seeking Approval on Different Fronts
 In mid-2006 Genmab announced the launch of a second pivotal phase III clinical trial of HuMax-CD20 as a single agent:
this time for treating follicular lymphoma patients who have failed chemoimmunotherapy combinations
incorporating Rituxan or Rituxan given as maintenance therapy. While the trial is interesting in itself as
expanding the options for follicular lymphoma patients, it also serves as another clinical platform to
demonstrate the possible superior efficacy of HuMax-CD20. The results of this
clinical trial should be of interest to CLL patients as well if it results in market entry for this drug. Read the details in
HuMax-CD20 Monotherapy in Refractory Follicular Lymphoma. (1/21/07)
In mid-2006 Genmab announced the launch of a second pivotal phase III clinical trial of HuMax-CD20 as a single agent:
this time for treating follicular lymphoma patients who have failed chemoimmunotherapy combinations
incorporating Rituxan or Rituxan given as maintenance therapy. While the trial is interesting in itself as
expanding the options for follicular lymphoma patients, it also serves as another clinical platform to
demonstrate the possible superior efficacy of HuMax-CD20. The results of this
clinical trial should be of interest to CLL patients as well if it results in market entry for this drug. Read the details in
HuMax-CD20 Monotherapy in Refractory Follicular Lymphoma. (1/21/07)
Chemoimmunotherapy
HuMax-CD20 plus Fludarabine plus Cyclophosphamide
New Phase II H+FC Clinical Trial
 Genmab has recently launched a chemoimmunotherapy combination clinical trial involving its anti-CD20 monoclonal
antibody, HuMax-CD20 (generic name ofatumumab). This
is a phase II dose comparison trial. The Genmab protocol combines HuMax-CD20 with fludarabine and
cyclophosphamide over a 6 month treatment cycle. The trial is currently being offered at four recruitment
centers in the United States. There is reason to hope that HuMax-CD20 will be more effective than Rituxan
both as a single agent as well as in chemoimmunotherapy combinations such as this. You can find our discussion
of this announcement at: HuMax+FC Announcement. (1/9/07)
Genmab has recently launched a chemoimmunotherapy combination clinical trial involving its anti-CD20 monoclonal
antibody, HuMax-CD20 (generic name ofatumumab). This
is a phase II dose comparison trial. The Genmab protocol combines HuMax-CD20 with fludarabine and
cyclophosphamide over a 6 month treatment cycle. The trial is currently being offered at four recruitment
centers in the United States. There is reason to hope that HuMax-CD20 will be more effective than Rituxan
both as a single agent as well as in chemoimmunotherapy combinations such as this. You can find our discussion
of this announcement at: HuMax+FC Announcement. (1/9/07)
Rituxan plus Fenretinide
A Match Made in Heaven?
A New Combination Therapy Is on Trial at the Hutch
 We announce the launch of a promising Phase II clinical trial at the Fred Hutchinson Cancer Research Center. The
trial combines Rituxan with a Vitamin A analogue called fenretinide, already in
wide use as a maintenance agent in other cancers. This combination offers the potential of low-toxicity and
possible synergy between the two agents — and that could mean an effective therapy for CLL. This trial
is sponsored and partly funded by CLL Topics — with your hard-earned donation dollars. That should tell
you what we think of this approach. Read and evaluate for yourself the logic behind the protocol and the
promise that it offers —
Rituxan plus Fenretinide: a Match Made in Heaven? (1/8/07)
We announce the launch of a promising Phase II clinical trial at the Fred Hutchinson Cancer Research Center. The
trial combines Rituxan with a Vitamin A analogue called fenretinide, already in
wide use as a maintenance agent in other cancers. This combination offers the potential of low-toxicity and
possible synergy between the two agents — and that could mean an effective therapy for CLL. This trial
is sponsored and partly funded by CLL Topics — with your hard-earned donation dollars. That should tell
you what we think of this approach. Read and evaluate for yourself the logic behind the protocol and the
promise that it offers —
Rituxan plus Fenretinide: a Match Made in Heaven? (1/8/07)
Almost Free Stem Cell Transplants
NIH Clinical Trial for Sibling Donor Transplants
A Unique Opportunity
 The National Institutes of Health in Bethesda, MD are conducting a clinical trial for sibling donor transplants that
may offer a unique therapeutic opportunity for CLL patients with aggressive disease and poor prognostics. In this
article, Stem Cell Transplants - Free of Charge
(Almost!), we review the background and rationale for this clinical trial, as well as give you contact
information. Potential participants both in the US and overseas should carefully consider this
unique opportunity that might otherwise be out of their reach for financial reasons. (1/3/07)
The National Institutes of Health in Bethesda, MD are conducting a clinical trial for sibling donor transplants that
may offer a unique therapeutic opportunity for CLL patients with aggressive disease and poor prognostics. In this
article, Stem Cell Transplants - Free of Charge
(Almost!), we review the background and rationale for this clinical trial, as well as give you contact
information. Potential participants both in the US and overseas should carefully consider this
unique opportunity that might otherwise be out of their reach for financial reasons. (1/3/07)
ASH 2025 Highlights
Summary of New Research Findings
Latest in Hematology
 The American Society of Hematology Annual meeting is the high point in the year for a variety of people dealing with
CLL: professional hematologists, research scientists and corporations in the pharmaceutical and biotech industries
— and to patients anxiously awaiting the next big development in therapy. Our team of reporters covering this
year's ASH annual meeting at Orlando, FL, describes the highlights of the 5-day meeting in this report:
ASH 2025 Highlights.
The American Society of Hematology Annual meeting is the high point in the year for a variety of people dealing with
CLL: professional hematologists, research scientists and corporations in the pharmaceutical and biotech industries
— and to patients anxiously awaiting the next big development in therapy. Our team of reporters covering this
year's ASH annual meeting at Orlando, FL, describes the highlights of the 5-day meeting in this report:
ASH 2025 Highlights.
Poor Prognostics
The Road to a Stem Cell Transplant
Greg's Story: A Cautionary Tale
 Greg's Story
is a case study based on the notes of an anonymous patient. It is a cautionary tale
about the dangers inherent in a relapse in the face of poor prognostics. Delaying
the decision to seek a mini-allo transplant, the only known cure at this point for poor
prognostic disease, can result in that choice being removed suddenly. You will
learn why Greg feels poor prognosis CLL should be taken very seriously and why a decision on pursuing a
mini-allo should be made early in the process.
Greg's Story
is a case study based on the notes of an anonymous patient. It is a cautionary tale
about the dangers inherent in a relapse in the face of poor prognostics. Delaying
the decision to seek a mini-allo transplant, the only known cure at this point for poor
prognostic disease, can result in that choice being removed suddenly. You will
learn why Greg feels poor prognosis CLL should be taken very seriously and why a decision on pursuing a
mini-allo should be made early in the process.
Tregs Revealed
The Body's Way of Controlling the Mayhem
Key Players in a Delicate Balancing Act
 The human immune system performs a delicate balancing act between safeguarding the body's own vital tissues and
aggressively destroying pathogens and cancers. In recent years research has revealed how a key role in this act
is performed by immune system cells called Tregs. We review the critical role of these important players in an
article titled Tregs Revealed. Understanding how these
cells function and their relationship to the rest of the immune system is central to designing effective
therapies for CLL — and to controlling the unwanted side effect of autoimmune disease.
The human immune system performs a delicate balancing act between safeguarding the body's own vital tissues and
aggressively destroying pathogens and cancers. In recent years research has revealed how a key role in this act
is performed by immune system cells called Tregs. We review the critical role of these important players in an
article titled Tregs Revealed. Understanding how these
cells function and their relationship to the rest of the immune system is central to designing effective
therapies for CLL — and to controlling the unwanted side effect of autoimmune disease.
Fundraising and Sponsorship Philosophy
How We Select Research Projects to Sponsor and Support
Factoring Patients' Priorities into Clinical Trials
 When it comes to spending our donors' money on clinical trials, we try to make every last dollar count. Even more
important, we try to get projects launched that have the potential to make a difference to patient survival and
quality of life. Please read
Our Fundraising and Sponsorship Philosophy to understand objectives and the logic behind our efforts.
When it comes to spending our donors' money on clinical trials, we try to make every last dollar count. Even more
important, we try to get projects launched that have the potential to make a difference to patient survival and
quality of life. Please read
Our Fundraising and Sponsorship Philosophy to understand objectives and the logic behind our efforts.
Infections: Who Is Most at Risk?
Prognostic Indicators Can Identify Infection Risk
Know Your Enemy
 Serious infections account for an alarming proportion of deaths among CLL patients.
But are some patients more at risk than others? What are the prognostic indicators
which can help you assess the risk of serious infections in your own case? In
Who Is Most at Risk?
we review a very recent article that looks at the different risk buckets and provides
some practical pointers to improve your chances of staying out of the hospital.
Serious infections account for an alarming proportion of deaths among CLL patients.
But are some patients more at risk than others? What are the prognostic indicators
which can help you assess the risk of serious infections in your own case? In
Who Is Most at Risk?
we review a very recent article that looks at the different risk buckets and provides
some practical pointers to improve your chances of staying out of the hospital.
Steroid-Campath Combinations
An Expert Looks at Therapies for p53 Defective CLL
A Sledgehammer To Be Used Wisely
 A defect in the p53 gene is a poor prognostic marker and predicts aggressive disease.
Patients who have this defect tend to respond poorly to standard chemotherapy.
Finding new and better ways of treating CLL patients with p53 defects is therefore a
major priority in CLL clinical research. In his article,
Steroid-Campath Combinations,
Dr. Andrew Pettitt of the Royal Liverpool University Hospital evaluates agents that have
promise in this area and addresses the important questions of when to treat such patients and
how far to go with the treatment.
A defect in the p53 gene is a poor prognostic marker and predicts aggressive disease.
Patients who have this defect tend to respond poorly to standard chemotherapy.
Finding new and better ways of treating CLL patients with p53 defects is therefore a
major priority in CLL clinical research. In his article,
Steroid-Campath Combinations,
Dr. Andrew Pettitt of the Royal Liverpool University Hospital evaluates agents that have
promise in this area and addresses the important questions of when to treat such patients and
how far to go with the treatment.
Sponsored Clinical Trial
CD20 Shaving with Rituxan
Are Standard Dosages Way Too Much?
 The original dosing scheme for Rituxan was quite arbitrary — but it seemed to do the job.
A research team at the University of Virginia is now examining the finer features of how Rituxan
works and is asking an important question: when does it make sense to use less
of this drug than the dosage standard? In this article we examine the concept of "CD20 shaving"
and introduce a research project at UVA that we will be sponsoring and supporting financially.
The answers from this research can have an important bearing on the best way to utilize this valuable
monoclonal antibody. Details in
CD20 Shaving with Rituxan.
The original dosing scheme for Rituxan was quite arbitrary — but it seemed to do the job.
A research team at the University of Virginia is now examining the finer features of how Rituxan
works and is asking an important question: when does it make sense to use less
of this drug than the dosage standard? In this article we examine the concept of "CD20 shaving"
and introduce a research project at UVA that we will be sponsoring and supporting financially.
The answers from this research can have an important bearing on the best way to utilize this valuable
monoclonal antibody. Details in
CD20 Shaving with Rituxan.
Super Bugs: Staph
MRSA Is on the Loose
The Dangers of Drug Resistant Bacterial Infections
 Recent reports prove that infections from strains of drug-resistant bacteria are far more prevalent than
previously thought, accounting for more than half of all skin infections treated in hospital emergency rooms.
Infectious diseases experts are trying hard to get the word out. While this poses a serious threat to public
health, CLL patients will do well to pay special attention to this phenomenon and take some precautions —
so that they may avoid these difficult and persistent infections which their systems are
ill-equipped to fight. Read about these super bugs in
The Dangers of Drug Resistant Bacterial Infections.
Recent reports prove that infections from strains of drug-resistant bacteria are far more prevalent than
previously thought, accounting for more than half of all skin infections treated in hospital emergency rooms.
Infectious diseases experts are trying hard to get the word out. While this poses a serious threat to public
health, CLL patients will do well to pay special attention to this phenomenon and take some precautions —
so that they may avoid these difficult and persistent infections which their systems are
ill-equipped to fight. Read about these super bugs in
The Dangers of Drug Resistant Bacterial Infections.
HuMax-CD20: Risks and Rewards
Belt, Suspenders and Cummerbund
Get to Know Ofatumumab
 HuMax-CD20 derives part of its improved efficacy from its capacity to work with a patient's complement
system to destroy B-cells. In HuMax-CD20
Risks and Rewards, we examine the potential risks of complement depletion that may accompany intensive
treatment with this monoclonal antibody — and suggest a possible precautionary measure for those who
are taking part in the Phase III trial of this agent for fludarabine- and Campath-refractory patients.
HuMax-CD20 derives part of its improved efficacy from its capacity to work with a patient's complement
system to destroy B-cells. In HuMax-CD20
Risks and Rewards, we examine the potential risks of complement depletion that may accompany intensive
treatment with this monoclonal antibody — and suggest a possible precautionary measure for those who
are taking part in the Phase III trial of this agent for fludarabine- and Campath-refractory patients.
HuMax-CD20
A Smarter Monoclonal on Trial
What Makes HuMax-CD20 Different from Rituxan?
 In this article we take a closer look at the new agent starring in Genmab's recently announced Phase III
clinical trial for fludarabine and Campath refractory CLL patients. This well-designed trial keys in on
HuMax-CD20's strengths which we hope will provide significantly better results than singe-agent Rituxan,
especially in this difficult patient cohort. To learn more about this pivotal trial and to understand how
this important new monoclonal antibody works, read
A Smarter Monoclonal on Trial.
In this article we take a closer look at the new agent starring in Genmab's recently announced Phase III
clinical trial for fludarabine and Campath refractory CLL patients. This well-designed trial keys in on
HuMax-CD20's strengths which we hope will provide significantly better results than singe-agent Rituxan,
especially in this difficult patient cohort. To learn more about this pivotal trial and to understand how
this important new monoclonal antibody works, read
A Smarter Monoclonal on Trial.
EBV
The Kiss that Can Kill
The Enemy Within
 The Epstein-Barr Virus (EBV) has long been known as a complicating factor in many cancers. Recent research has shown
just how serious is the threat posed by this virus in CLL. EBV is estimated to be present in over 90% of adults in
the western world but is normally kept in dormancy by active T-cells. Read
The Enemy Within to learn how EBV can interfere
with normal T-cell function and how T-cell damaging therapy with agents such as fludarabine and Campath can unleash
EBV with life-threatening consequences. We also discuss how patients may be able to use a common statin to improve
their defenses against this virus.
The Epstein-Barr Virus (EBV) has long been known as a complicating factor in many cancers. Recent research has shown
just how serious is the threat posed by this virus in CLL. EBV is estimated to be present in over 90% of adults in
the western world but is normally kept in dormancy by active T-cells. Read
The Enemy Within to learn how EBV can interfere
with normal T-cell function and how T-cell damaging therapy with agents such as fludarabine and Campath can unleash
EBV with life-threatening consequences. We also discuss how patients may be able to use a common statin to improve
their defenses against this virus.
Infectious Complications
Best Practices
How to Treat and Manage Infections in CLL
 Infectious complications are the major cause of mortality in CLL and affect the quality of life of most patients.
The best practices in managing infections have changed over time, tracking our knowledge of the molecular mechanisms
at work and reflecting the nature of the therapies used to treat the CLL itself. In our article
Infectious Complications of CLL,
we review the latest expert findings. We also highlight the need for patients to be actively involved in discussing
these best practices with their local oncologists, to ensure that they receive appropriate care.
Infectious complications are the major cause of mortality in CLL and affect the quality of life of most patients.
The best practices in managing infections have changed over time, tracking our knowledge of the molecular mechanisms
at work and reflecting the nature of the therapies used to treat the CLL itself. In our article
Infectious Complications of CLL,
we review the latest expert findings. We also highlight the need for patients to be actively involved in discussing
these best practices with their local oncologists, to ensure that they receive appropriate care.
Caution on EPO
The Dark Side of Epoetin - Getting Darker
Be Careful What You Ask For !
 The results of a new meta study have been published in the Journal of the National Cancer Institute.
The authors find additional reasons to question the usage of high doses of epoetin drugs to achieve
"normal" levels of hemoglobin. Biases in earlier studies may have understated the problems.
We present our review in
The Dark Side of EPO - Getting Darker.
The results of a new meta study have been published in the Journal of the National Cancer Institute.
The authors find additional reasons to question the usage of high doses of epoetin drugs to achieve
"normal" levels of hemoglobin. Biases in earlier studies may have understated the problems.
We present our review in
The Dark Side of EPO - Getting Darker.
Genmab in the News
HuMax-CD20 in Action
Harvey is Back!
 We have an opportunity to see HuMax-CD20 in action when Harvey and Serena pack their bags and travel to England
in pursuit of therapy with this new agent. To learn more, read:
Genmab in the News.
We have an opportunity to see HuMax-CD20 in action when Harvey and Serena pack their bags and travel to England
in pursuit of therapy with this new agent. To learn more, read:
Genmab in the News.
Sponsored Clinical Trial
Low Hanging Fruit
Improving Routine Immunizations
 Sometimes, when luck is running our way and all the stars are in their correct positions in
the heavens, everything comes together just so and we have an opportunity to do ourselves a favor as a community.
In our new article, Improving Immunizations, we present
a clinical trial to test a method that may boost the efficacy of routine immunizations for CLL patients. We are
happy to report that Prof. Terry Hamblin is now working on making this trial a reality.
Sometimes, when luck is running our way and all the stars are in their correct positions in
the heavens, everything comes together just so and we have an opportunity to do ourselves a favor as a community.
In our new article, Improving Immunizations, we present
a clinical trial to test a method that may boost the efficacy of routine immunizations for CLL patients. We are
happy to report that Prof. Terry Hamblin is now working on making this trial a reality.
Chemotherapy
A Direct Comparison of F+C vs F
Is More Necessarily Better?
 The latest issue of Blood carries an article from the German CLL
Study Group comparing single-agent fludarabine against a
combination of fludarabine plus cyclophosphamide. This is a
well-structured, rigorous study and the study group comes to
some surprising conclusions. Read
F+C vs F for our analysis of the
study and comments from an editorial in Blood by Dr. Neil Kay.
The latest issue of Blood carries an article from the German CLL
Study Group comparing single-agent fludarabine against a
combination of fludarabine plus cyclophosphamide. This is a
well-structured, rigorous study and the study group comes to
some surprising conclusions. Read
F+C vs F for our analysis of the
study and comments from an editorial in Blood by Dr. Neil Kay.
Vaccines
A Vaccine Trial for CLL Patients
Finally, It's Our Turn ...
 Genitope Corp. has announced a clinical trial of their MyVax proprietary
vaccine technology for untreated, early stage CLL patients. We
examine this promising technology and the structure of the
Genitope clinical trial in our new article,
A Vaccine Trial for CLL Patients.
Genitope Corp. has announced a clinical trial of their MyVax proprietary
vaccine technology for untreated, early stage CLL patients. We
examine this promising technology and the structure of the
Genitope clinical trial in our new article,
A Vaccine Trial for CLL Patients.
Case Studies
Forest Bump on His Experience in FCR Lite Trial
The Road Turns Less Bumpy for Our Hero
 Our intrepid volunteer, thinly disguised as "Forest Bump", reports
on his experience as a participant in the "FCR Lite" clinical trial being
conducted at the University of Pittsburgh Cancer Institute.
Forest, you will discover, has a unique personality and view of
life. We hope his tale,
Forest Bump, will be useful to those interested in a
"lighter" version of the widely used FCR protocol.
Our intrepid volunteer, thinly disguised as "Forest Bump", reports
on his experience as a participant in the "FCR Lite" clinical trial being
conducted at the University of Pittsburgh Cancer Institute.
Forest, you will discover, has a unique personality and view of
life. We hope his tale,
Forest Bump, will be useful to those interested in a
"lighter" version of the widely used FCR protocol.
Rituxan Therapy
Update on New Adverse Effects netticasino
Harvey Runs into a Road-block
 Since our hypothetical patient Harvey has developed a modest following of his own,
our readers will be interested in some new developments.
Harvey’s experience coincides with recently published reports on
new adverse effects of Rituxan therapy.
While the post-marketing surveillance of Rituxan has so far identified only a small percentage of patients
having these serious adverse effects, the danger is high and very real for these patients
and therefore worth discussing. Find the details in our next installment of the Harvey Saga,
Harvey Runs into a Road-block.
Since our hypothetical patient Harvey has developed a modest following of his own,
our readers will be interested in some new developments.
Harvey’s experience coincides with recently published reports on
new adverse effects of Rituxan therapy.
While the post-marketing surveillance of Rituxan has so far identified only a small percentage of patients
having these serious adverse effects, the danger is high and very real for these patients
and therefore worth discussing. Find the details in our next installment of the Harvey Saga,
Harvey Runs into a Road-block.
Therapy Choices
The Price of Collateral Damage
Winning the Battle but Losing the War?
 Treating CLL needs a different approach from treating more familiar solid
cancers. In this article we discuss the costs and benefits of
different strategies. If you are considering therapy this
article may help you understand your options a little better:
Winning the Battle but Losing the War?
Treating CLL needs a different approach from treating more familiar solid
cancers. In this article we discuss the costs and benefits of
different strategies. If you are considering therapy this
article may help you understand your options a little better:
Winning the Battle but Losing the War?
Phytochemicals
The Latest on Green Tea
Details Make a Difference
 We report on some recent information that has come to light on green tea
extracts and EGCG in cancer treatment and prevention and
provide an update on Project Alpha. Read the details in
Latest on Green Tea.
We report on some recent information that has come to light on green tea
extracts and EGCG in cancer treatment and prevention and
provide an update on Project Alpha. Read the details in
Latest on Green Tea.
Clinical Trial Updates
The Search for Consistency in Reporting Results
Comparing Apples to Kumquats and One Small Victory
 We revisit two clinical trials discussed in past articles - FCR and
PCR. Comparing the results of these two trials is not easy but we nevertheless
make a brave attempt. We also discuss new trials in the offing
and report a welcome change in the design of the FCR + M
clinical trial. Sometimes it helps to tilt with windmills! Read
all about these things in Clinical Trial Updates.
We revisit two clinical trials discussed in past articles - FCR and
PCR. Comparing the results of these two trials is not easy but we nevertheless
make a brave attempt. We also discuss new trials in the offing
and report a welcome change in the design of the FCR + M
clinical trial. Sometimes it helps to tilt with windmills! Read
all about these things in Clinical Trial Updates.
FCR 'Lite'
The Search for Efficacy with Lower Toxicity
New Clinical Trial Seeks the Goldilocks Solution
 A new chemoimmunotherapy combination employing a higher dosage of Rituxan with
reduced dosages of fludarabine and cyclophosphamide, is
being tested in a clinical trial at the University of Pittsburgh
Cancer Institute. The investigators would like to achieve high
response rates with lower toxicity compared to FCR "Classic"
pioneered at M. D. Anderson Cancer Center. We review the
protocol and comment on the clinical trial design in our article
on FCR Lite. We also
interviewed Dr. Kenneth Foon, the principal investigator for
this clinical trial at UPMC.
A new chemoimmunotherapy combination employing a higher dosage of Rituxan with
reduced dosages of fludarabine and cyclophosphamide, is
being tested in a clinical trial at the University of Pittsburgh
Cancer Institute. The investigators would like to achieve high
response rates with lower toxicity compared to FCR "Classic"
pioneered at M. D. Anderson Cancer Center. We review the
protocol and comment on the clinical trial design in our article
on FCR Lite. We also
interviewed Dr. Kenneth Foon, the principal investigator for
this clinical trial at UPMC.
RF Risks & Benefits
High Risk Groups Relapse Sooner online casino
Further Analysis from Ohio State
 As patients it is important to us that our remissions be long and
that we maximize survival. These objectives are not always
captured by response statistics typically reported in
cutting-edge phase II clinical trials of new agents. In our
new article on RF Therapy, we
focus on a follow-up paper published by Drs. John Byrd, et. al.,
that goes straight to the point – CRs and PRs don't mean the
same thing to different prognostic risk groups, unlike the
proverbial rose that smells just as sweet to anyone who cares
to sniff.
As patients it is important to us that our remissions be long and
that we maximize survival. These objectives are not always
captured by response statistics typically reported in
cutting-edge phase II clinical trials of new agents. In our
new article on RF Therapy, we
focus on a follow-up paper published by Drs. John Byrd, et. al.,
that goes straight to the point – CRs and PRs don't mean the
same thing to different prognostic risk groups, unlike the
proverbial rose that smells just as sweet to anyone who cares
to sniff.
HuMax Clinical Trial
Early Results from GenMab's Anti-CD20 Monoclonal Antibody
The Half-Full Glass
 We have high hopes for the new generation of monoclonal antibodies
that are under development and have discussed new trials under
way to test these agents in the clinic. Early results form one
such trial, involving GenMab's HuMax, were highlighted at the
2005 ASH conference in Atlanta. We offer our analysis of the
published results in Results from
GenMab's HuMax Clinical Trial.
We have high hopes for the new generation of monoclonal antibodies
that are under development and have discussed new trials under
way to test these agents in the clinic. Early results form one
such trial, involving GenMab's HuMax, were highlighted at the
2005 ASH conference in Atlanta. We offer our analysis of the
published results in Results from
GenMab's HuMax Clinical Trial.
Avian Flu and CLL
Recognizing Vulnerabilities and Taking Precautions
A Cave Is No Place to Live
 You cannot have missed all the references to "bird flu", "avian
influenza", "H5N1" and "pandemic flu" in the mainstream media.
There are as many opinions about bird flu as there are chickens
in China. We would like to add our two cents, but strictly focused on issues
as they pertain to CLL patients,
the special risks we face as immune compromised individuals. We
also attach some up-to-date reference material that you might
find useful, especially when you talk to your doctors about your
needs. Read our article on
Avian Flu and CLL.
You cannot have missed all the references to "bird flu", "avian
influenza", "H5N1" and "pandemic flu" in the mainstream media.
There are as many opinions about bird flu as there are chickens
in China. We would like to add our two cents, but strictly focused on issues
as they pertain to CLL patients,
the special risks we face as immune compromised individuals. We
also attach some up-to-date reference material that you might
find useful, especially when you talk to your doctors about your
needs. Read our article on
Avian Flu and CLL.
MCL - a Wolf in CLL Clothing
Accurate Diagnosis Leads to Better Treatment
Know the Name of Your Enemy
 FISH analysis is one of the key prognostic tests used to identify the
type of CLL a patient has. However, the usual FISH panels
contain probes for only the most common cytogenetic
abnormalities. Rarer aberrations all get lumped together in a
basket labeled "normal". In this article,
MCL - a Wolf in CLL Clothing, we
examine how a probe for one such rare aberration, the "t(11:14)"
translocation, can yield valuable information in distinguishing
CLL from the more aggressive Mantle Cell Lymphoma, for which
treatment choices are not the same. Having this additional probe
included in your FISH test may well depend upon your negotiating
skills.
FISH analysis is one of the key prognostic tests used to identify the
type of CLL a patient has. However, the usual FISH panels
contain probes for only the most common cytogenetic
abnormalities. Rarer aberrations all get lumped together in a
basket labeled "normal". In this article,
MCL - a Wolf in CLL Clothing, we
examine how a probe for one such rare aberration, the "t(11:14)"
translocation, can yield valuable information in distinguishing
CLL from the more aggressive Mantle Cell Lymphoma, for which
treatment choices are not the same. Having this additional probe
included in your FISH test may well depend upon your negotiating
skills.
Targeting Bcl-2
Anti-death Protein Protects Cancer Cells
We Review Clinical Trials of Three Different Drugs
 Clinical trials are currently under way testing three different drugs to
suppress or attack a family of proteins that confers longevity
to CLL cells. We review the science behind this approach — and
give you our take on the drugs and the clinical trials — in
Targeting Bcl-2.
Clinical trials are currently under way testing three different drugs to
suppress or attack a family of proteins that confers longevity
to CLL cells. We review the science behind this approach — and
give you our take on the drugs and the clinical trials — in
Targeting Bcl-2.
Flu Preparedness
Pneumonia Is the Single Biggest Killer of CLL Patients
Some Pointers to Resources and Education
 The annual influenza season will soon be upon us. In
Flu Preparedness we offer
some research findings and practical advice on making it through
this season, whether we are faced with the common or garden
variety of flu - or a more ominous humanized version of the
avian flu.
The annual influenza season will soon be upon us. In
Flu Preparedness we offer
some research findings and practical advice on making it through
this season, whether we are faced with the common or garden
variety of flu - or a more ominous humanized version of the
avian flu.
T-Cell Therapy
Clinical Trial at the Hutch Now to Recruit CLL Patients
Trained CD-20 Targeted Killer T-Cells to the Rescue
 Another interesting clinical trial will soon be open to CLL patients.
T-cells collected from patients are grown into large armies and
targeted to kill only cells with the CD-20 marker. These
activated and targeted killer T-cells will be used in the
clean-up of minimum residual disease left after more traditional
therapies. This innovative approach is the basis for a Phase I
clinical trial at the Fred Hutchinson Cancer Research Center in
Seattle. Read our review of this latest evolution in using CTLs
in
Autologous CD-20 Targeted T-Cell Therapy.
Another interesting clinical trial will soon be open to CLL patients.
T-cells collected from patients are grown into large armies and
targeted to kill only cells with the CD-20 marker. These
activated and targeted killer T-cells will be used in the
clean-up of minimum residual disease left after more traditional
therapies. This innovative approach is the basis for a Phase I
clinical trial at the Fred Hutchinson Cancer Research Center in
Seattle. Read our review of this latest evolution in using CTLs
in
Autologous CD-20 Targeted T-Cell Therapy.
Clinical Trials
A Patient Sponsored Clinical Trial
Project Alpha Kickoff
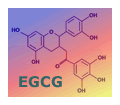 It has taken longer than we thought it would, but we are
pleased to announce Project Alpha is finally ready to start
recruiting patients. It represents a first of its kind
collaboration between patients and the research community.
Your hard earned money went into making this "EGCG trial" a
reality. You can read all the details of this newly
announced clinical trial at Mayo Clinic, Rochester MN, in
our new article,
Project Alpha Kickoff.
It has taken longer than we thought it would, but we are
pleased to announce Project Alpha is finally ready to start
recruiting patients. It represents a first of its kind
collaboration between patients and the research community.
Your hard earned money went into making this "EGCG trial" a
reality. You can read all the details of this newly
announced clinical trial at Mayo Clinic, Rochester MN, in
our new article,
Project Alpha Kickoff.
Case Studies
A Few Short Years Can Change Some Important Things
Death by Conventional Wisdom
 The conventional wisdom of a few years ago can prove dangerous when doctors treat
patients by outdated rules of thumb. In our (2005) critique of this case study reported
by a professional journal in 1999, we identify the necessity for both physicians
and patients to understand and employ modern risk-based strategies in dealing with
this varied disease. Read "
Death by Conventional Wisdom" to understand why staying on top of new developments
is critical for your doctor — and even more so for you.
The conventional wisdom of a few years ago can prove dangerous when doctors treat
patients by outdated rules of thumb. In our (2005) critique of this case study reported
by a professional journal in 1999, we identify the necessity for both physicians
and patients to understand and employ modern risk-based strategies in dealing with
this varied disease. Read "
Death by Conventional Wisdom" to understand why staying on top of new developments
is critical for your doctor — and even more so for you.
Risk-Adapted Strategies
Experts Summarize the State of CLL Therapy
Where Are We?
 Drawing from a group of top-rated experts, Dr Marco Montillo in Milan
has produced an overview of where we are in understanding and treating CLL.
Patients and their oncologists should pay close attention to the historical
evidence and conclusions presented by this group. Read about it in
Risk-adapted Strategies.
Drawing from a group of top-rated experts, Dr Marco Montillo in Milan
has produced an overview of where we are in understanding and treating CLL.
Patients and their oncologists should pay close attention to the historical
evidence and conclusions presented by this group. Read about it in
Risk-adapted Strategies.
Graft versus Host Disease
Controlling this Killer is the Holy Grail of Transplants
GVHD in Allogeneic Transplants
 Graft-versus-host-disease (GVHD) is an example of systemic and potentially
life threatening inflammation. It is one of the major causes of death in
stem cell transplants. Controlling GVHD is the holy grail of all transplant
researchers. This article discusses the causes
GVHD, and some commonsense things you can do to protect yourself.
Graft-versus-host-disease (GVHD) is an example of systemic and potentially
life threatening inflammation. It is one of the major causes of death in
stem cell transplants. Controlling GVHD is the holy grail of all transplant
researchers. This article discusses the causes
GVHD, and some commonsense things you can do to protect yourself.
Stem Cell Transplants
The Only Real Cure Out There, for Now
New Papers Spell Out the Chances for Success
 As the years go by, survival statistics from stem cell transplants performed
in the past have been accumulating. We review the surprising results reported
in two newly published papers from the Hutch in our article,
The Only Real Cure Out There, for Now.
As the years go by, survival statistics from stem cell transplants performed
in the past have been accumulating. We review the surprising results reported
in two newly published papers from the Hutch in our article,
The Only Real Cure Out There, for Now.
Chemotherapy Trials
Hsp 90 and Its Inhibition by 17-AAG
An Important New Trial Has Been Announced
 17-AAG may turn out to be an important therapeutic drug for CLL.
It has an interesting mechanism of action and should in theory work
well in poor prognostic cases. An important new phase I trial of
17-AAG has been announced and is recruiting patients at OSU, soon to
be followed by Dana Farber. You can read all about it in our article
17-AAG - An Important New
Trial Has Been Announced.
17-AAG may turn out to be an important therapeutic drug for CLL.
It has an interesting mechanism of action and should in theory work
well in poor prognostic cases. An important new phase I trial of
17-AAG has been announced and is recruiting patients at OSU, soon to
be followed by Dana Farber. You can read all about it in our article
17-AAG - An Important New
Trial Has Been Announced.
Chemotherapy
Latest on Candidate 17-AAG
Hope Springs Eternal
 The patient community is ever hopeful about new drug candidates
that appear to have potential in the lab. In
17-AAG
we examine a new
agent that is receiving attention, 17-AAG, which is on its way to starring in a clinical trial at Ohio State under the orchestration of Dr. John Byrd.
The patient community is ever hopeful about new drug candidates
that appear to have potential in the lab. In
17-AAG
we examine a new
agent that is receiving attention, 17-AAG, which is on its way to starring in a clinical trial at Ohio State under the orchestration of Dr. John Byrd.
Combination Immunotherapy
Campath plus G-CSF Can Be Dangerous
A Scary Story: Telling It Like It Is
 The combination of Campath and G-CSF appears logical enough as
an approach to reaping the benefits of Campath therapy while
keeping the immune suppression and infections typically associated
with it under control. However, the co-administration of G-CSF
with intravenous Campath to patients enrolled in a phase II trial
at Ohio State produced unexpected and discouraging results. The
prompt publication of the results is a credit to the Ohio State team.
It is important that we understand the results of this trial as we
consider our therapy alternatives. Read about it in
Campath plus G-CSF.
The combination of Campath and G-CSF appears logical enough as
an approach to reaping the benefits of Campath therapy while
keeping the immune suppression and infections typically associated
with it under control. However, the co-administration of G-CSF
with intravenous Campath to patients enrolled in a phase II trial
at Ohio State produced unexpected and discouraging results. The
prompt publication of the results is a credit to the Ohio State team.
It is important that we understand the results of this trial as we
consider our therapy alternatives. Read about it in
Campath plus G-CSF.
Therapy Choices
Restoring Humoral Functions in B-CLL
Possible Benefits of IVIg
 Most CLL patients have reduced immunoglobulin (Ig),
the levels dropping gradually over time. Lack of
proper B-cell function in generating a wide repertoire
of Ig needed is one of the reasons why CLL patients get
so many infections. This article,
Possible Benefits
of IVIg, describes what happens when B-cell function
is compromised by cancer or therapies used to control the
cancer, causing sub-normal Ig levels. One possible solution
for this defect is, of course, IVIg. Recent research
suggests there may be even more important and unexpected
benefits to be gained from IVIg.
Most CLL patients have reduced immunoglobulin (Ig),
the levels dropping gradually over time. Lack of
proper B-cell function in generating a wide repertoire
of Ig needed is one of the reasons why CLL patients get
so many infections. This article,
Possible Benefits
of IVIg, describes what happens when B-cell function
is compromised by cancer or therapies used to control the
cancer, causing sub-normal Ig levels. One possible solution
for this defect is, of course, IVIg. Recent research
suggests there may be even more important and unexpected
benefits to be gained from IVIg.
Prognostic Indicators
Prognostic Test Packages are Now Available
Quest Diagnostics Is First to the Table
 Your doctor can now order a package of CLL-specific prognostic
tests from the largest company in the medical test industry,
Quest Diagnostics, Inc. Doctors can now get the tests done as
a package deal - and at a reasonable cost to the patient.
It has been our crusade to take out some of the road blocks
to make prognostic testing accessible to local healthcare
providers who guide the therapy choices of their CLL patients
every day. Read about this important and very useful development
in our article on
Prognostic and Monitoring Tests. We provide all the details
of how to get the tests done and the logic you may need to convince
your doctor to order them.
Your doctor can now order a package of CLL-specific prognostic
tests from the largest company in the medical test industry,
Quest Diagnostics, Inc. Doctors can now get the tests done as
a package deal - and at a reasonable cost to the patient.
It has been our crusade to take out some of the road blocks
to make prognostic testing accessible to local healthcare
providers who guide the therapy choices of their CLL patients
every day. Read about this important and very useful development
in our article on
Prognostic and Monitoring Tests. We provide all the details
of how to get the tests done and the logic you may need to convince
your doctor to order them.
New Clinical Trial at Mayo
Protocol for Previously Untreated Patients
Short Duration Campath plus Rituxan
 A new protocol combining short duration subcutaneous Campath
with standard dose Rituxan has been launched at the Mayo Clinic,
Rochester, MN. It targets previously untreated Rai Stage 0-II
patients. Read our review of the protocol and the relevant detail
and background in
Clinical Trial to Test Rituxan plus Short Duration Campath.
A new protocol combining short duration subcutaneous Campath
with standard dose Rituxan has been launched at the Mayo Clinic,
Rochester, MN. It targets previously untreated Rai Stage 0-II
patients. Read our review of the protocol and the relevant detail
and background in
Clinical Trial to Test Rituxan plus Short Duration Campath.
Campath Therapy
Good News for the Tough Cases
The Half-full Glass
 Another piece of solid clinical research with important implications
has just been published online in the Journal of Clinical Oncology. In it, Paul Moreton, et al., report on the results
of a 7-year phase II study involving late stage and refractory patients.
The British team concludes that eradication of minimum residual disease
with Campath has a surprisingly good effect on survival in this cohort
of tough cases. We review the Moreton article and offer our analysis in
Good News for the Tough Cases.
Another piece of solid clinical research with important implications
has just been published online in the Journal of Clinical Oncology. In it, Paul Moreton, et al., report on the results
of a 7-year phase II study involving late stage and refractory patients.
The British team concludes that eradication of minimum residual disease
with Campath has a surprisingly good effect on survival in this cohort
of tough cases. We review the Moreton article and offer our analysis in
Good News for the Tough Cases.
Staging is Superseded
The New Think on CLL Is Finally Here csgo case
The Dawn of a New Era
 An important new review article has been published
in the New England Journal of Medicine. The Rai/Binet
staging systems have been important in triggering
therapy decisions. In this article the authors, who
include Dr. Kanti Rai himself, bluntly state that the
staging system with its emphasis on watch & wait
should be replaced by better methods using modern
prognostic indicators. We cover this important article
as well as several other related works in
The Dawn of a New Era.
An important new review article has been published
in the New England Journal of Medicine. The Rai/Binet
staging systems have been important in triggering
therapy decisions. In this article the authors, who
include Dr. Kanti Rai himself, bluntly state that the
staging system with its emphasis on watch & wait
should be replaced by better methods using modern
prognostic indicators. We cover this important article
as well as several other related works in
The Dawn of a New Era.
Prognostic Indicators
Progress in Getting the Tests Made Accessible
Our Quest For Solutions
 The value of getting the right prognostic tests done is
slowly gaining acceptance in the patient community. However,
the practical difficulties involved are numerous and each
patient has to deal with his or her providers as well as
he or she can. In this article,
Progress on Prognostics, we review our own progress in de-tangling
the logistical and communication nightmare for the patient community.
In CLL, as in life, what you get is what you negotiate - and we have been busy.
The value of getting the right prognostic tests done is
slowly gaining acceptance in the patient community. However,
the practical difficulties involved are numerous and each
patient has to deal with his or her providers as well as
he or she can. In this article,
Progress on Prognostics, we review our own progress in de-tangling
the logistical and communication nightmare for the patient community.
In CLL, as in life, what you get is what you negotiate - and we have been busy.
Physicians' Corner
Progress in Finding a Cure
Are We There Yet?
 Professor Terry Hamblin needs no introduction to CLL patients.
His seminal work on the prognostic value of the IgVH gene mutation
status has become a cornerstone in the risk stratification of CLL
patients. In his article,
Are We There Yet?, Dr. Hamblin discusses criteria for initiating
treatment as well as what treatment can and cannot do for the
patient. He goes on to review the current status of combination
chemo-immunotherapy clinical trials. Read this article for a candid
assessment of where we are in the campaign to cure CLL.
Professor Terry Hamblin needs no introduction to CLL patients.
His seminal work on the prognostic value of the IgVH gene mutation
status has become a cornerstone in the risk stratification of CLL
patients. In his article,
Are We There Yet?, Dr. Hamblin discusses criteria for initiating
treatment as well as what treatment can and cannot do for the
patient. He goes on to review the current status of combination
chemo-immunotherapy clinical trials. Read this article for a candid
assessment of where we are in the campaign to cure CLL.
Nutrition and Chemoprevention
Omega-3 Fatty Acids free slots
A Fishy Tale
 Much current research is focused on the role played
by polyunsaturated fatty acids (PUFAs) in a very
broad range of cellular functions in humans. Our
modern diet and agricultural technology have created
a substantial imbalance between the two major groups
PUFAs that we consume. Restoring that balance is
critical to our ability to avoid and fight cancer
and other diseases. In
Omega-3 Fatty Acids we review current scientific
thought on the importance of increasing our consumption
of Omega-3 oils, derived mainly from fish.
Much current research is focused on the role played
by polyunsaturated fatty acids (PUFAs) in a very
broad range of cellular functions in humans. Our
modern diet and agricultural technology have created
a substantial imbalance between the two major groups
PUFAs that we consume. Restoring that balance is
critical to our ability to avoid and fight cancer
and other diseases. In
Omega-3 Fatty Acids we review current scientific
thought on the importance of increasing our consumption
of Omega-3 oils, derived mainly from fish.
CLL Complications
Oral Mucositis Can Be Deadly best online casino uk
A Real Pain in the Mouth
 Many CLL patients experience painful sores and infections
in their mouths, especially when they go through chemotherapy.
This incidence of 'oral mucositis' can have serious consequences
to the quality of life and even survival of these patients. There
may be a few things you can do to help yourself - read about them in
A Real Pain in the Mouth.
Many CLL patients experience painful sores and infections
in their mouths, especially when they go through chemotherapy.
This incidence of 'oral mucositis' can have serious consequences
to the quality of life and even survival of these patients. There
may be a few things you can do to help yourself - read about them in
A Real Pain in the Mouth.
Matching Made Simple
Is a Bone Marrow Transplant in Your Future?
Allogeneic Stem Cell Transplants Work Better with Closer Matches
 HLA matching of the potential donor with the recipient is
the first step in allogeneic stem cell transplants, whether
the stem cells are obtained from an adult donor or from
umbilical cord blood. With increasing frequency, stem
cell transplantation is viewed as an early alternative
for those with aggressive disease or high risk profiles,
especially for younger patients. Familiarity with the
concept of tissue typing will be useful as you embark upon
the transplant journey. To take some of the mystery out of
this process we present
Matching Made Simple.
HLA matching of the potential donor with the recipient is
the first step in allogeneic stem cell transplants, whether
the stem cells are obtained from an adult donor or from
umbilical cord blood. With increasing frequency, stem
cell transplantation is viewed as an early alternative
for those with aggressive disease or high risk profiles,
especially for younger patients. Familiarity with the
concept of tissue typing will be useful as you embark upon
the transplant journey. To take some of the mystery out of
this process we present
Matching Made Simple.
Monoclonal Antibodies
Sons of Rituxan and Campath
A New Generation of More Effective Antibodies Makes It into the Clinic
 The monoclonal antibodies Rituxan and Campath have changed
the landscape for CLL and a whole range of hematological
conditions. New improved versions of these monoclonal
antibodies are now coming out of the lab and into the
clinic. In our article
Sons of Rituxan and Campath, we examine the mechanisms
by which these agents achieve their effects - and the
methods for increasing their efficacy. Read about a new
CD20 monoclonal antibody, HuMax from GenMab, now in clinical
trials in Europe and in the US and, incidentally, satisfy
your curiosity about why we have a picture of a lobster
gracing this paragraph.
The monoclonal antibodies Rituxan and Campath have changed
the landscape for CLL and a whole range of hematological
conditions. New improved versions of these monoclonal
antibodies are now coming out of the lab and into the
clinic. In our article
Sons of Rituxan and Campath, we examine the mechanisms
by which these agents achieve their effects - and the
methods for increasing their efficacy. Read about a new
CD20 monoclonal antibody, HuMax from GenMab, now in clinical
trials in Europe and in the US and, incidentally, satisfy
your curiosity about why we have a picture of a lobster
gracing this paragraph.
Epigenetics
Genes Sleeping on the Job
New Therapies Based on a Better Understanding of Cell Biology
 An interesting new clinical trial has opened up for CLL patients,
one that may have potential for low toxicity as well as good
efficacy. As a refreshing change from the usual heavy doses of
chemotherapy, this trial is based on using very low doses of
drugs, often as little as ten times lower than the amounts used
under standard regimens. The rationale for this approach is
equally interesting. The DNA in a cell's nucleus is a vast
library of information which normally defines and regulates
how the cell functions. In cancer cells, however, the information
retrieval mechanism is often at fault. The idea here is to use
just enough drugs to correct this fault, and thereby allow the
cancer cell to kill itself. Click here to read
Epigenetics.
An interesting new clinical trial has opened up for CLL patients,
one that may have potential for low toxicity as well as good
efficacy. As a refreshing change from the usual heavy doses of
chemotherapy, this trial is based on using very low doses of
drugs, often as little as ten times lower than the amounts used
under standard regimens. The rationale for this approach is
equally interesting. The DNA in a cell's nucleus is a vast
library of information which normally defines and regulates
how the cell functions. In cancer cells, however, the information
retrieval mechanism is often at fault. The idea here is to use
just enough drugs to correct this fault, and thereby allow the
cancer cell to kill itself. Click here to read
Epigenetics.
Therapy Choices
Trust but Verify
Shopping for the Therapy that Is Right for You
 Picking your therapy based on the results reported from clinical
trials is not easy. You have to read the results carefully -
the devil is indeed in the detail. Comparing
trial results is only valid if the patient groups are well
matched between the trials - and sometimes the focus is on
the wrong result statistic. Unbiased, timely and complete
reporting of the results of human clinical trials is critical
to understanding the therapies out there - and your therapy
choices. Click here to read
Shopping for
the Therapy that is Right for You.
Picking your therapy based on the results reported from clinical
trials is not easy. You have to read the results carefully -
the devil is indeed in the detail. Comparing
trial results is only valid if the patient groups are well
matched between the trials - and sometimes the focus is on
the wrong result statistic. Unbiased, timely and complete
reporting of the results of human clinical trials is critical
to understanding the therapies out there - and your therapy
choices. Click here to read
Shopping for
the Therapy that is Right for You.

START RIGHT HERE:
► Read A CLL Primer
► Discover What Type of CLL you have;
► Read through our Topics Alerts Archive;
► Organize and maintain Your Charts;
► Get to know the recommended Best Practices for dealing with CLL;
► Become an informed customer before you go Shopping for Therapies or sign up for a clinical trial.
We have an online journal detailing the background and stages of a cord blood stem cell transplant for our case study patient, Harvey. Please do visit.
Clinical Research
Sponsored by
CLL Topics
Ever wonder what we do with the money we raise from your donations? cassino online
You can read about the clinical trials we have sponsored by clicking here: CLL Topics Sponsored Clinical Trials.
We explain the thinking behind these projects in an article titled Our Sponsorship Philosophy.

Open Clinical Trials
Editorial from the New England Journal of Medicine
October 3, 2025
“ … It is time for participants in clinical trials to take the initiative. People interested in participating in trials should consider only studies whose sponsors have fully registered them in an appropriate public database and agreed to publish their results.”
We agree. The full editorial is available in pdf form at the NEJM website. Patients are encouraged to read the editorial and understand the issues involved since they are critically important for individual participants in clinical trials as well as for the entire patient community.

Thanks to dedicated clinical researchers and brave volunteers enlisting in clinical trials, diagnostic tools and therapy options are changing fast in the treatment of CLL. We hope to assist you in understanding the type of CLL you have, the therapy options out there and some of the basic considerations in choosing how to proceed. The expression “living to fight another day” gains a special meaning when the whole ball game may change tomorrow.
Here are some of the therapies and agents we have discussed in our past articles:
Please do remember that the writers of the articles on CLLTopics.org are not medical professionals and do not provide medical advice or recommendations for treatment. Please consult and follow the advice of your own healthcare providers.

Fundraising
Status of Fundraising

We have reached our fundraising goal for 2025 and as of July 1, 2025 we are temporarily suspending our fundraising efforts. You can look up where we are in our fundraising both on the Donation Status page and the Acknowledgements page on this site. We are proud to acknowledge the generous gifts of many who support us in our work.
Managing Infections
In our Reference section you will find a valuable new addition on managing infections in the immune compromised patient. There is extensive material of particular relevance to CLL patients.
Patients' Corner
In our section called Patients' Corner, you can read other patients' opinions on diverse topics.
Physicians' Corner
In our section called Physicians' Corner, you will find articles and correspondence from physicians, researchers and other experts. Dr. Andrew Pettitt of the Royal Liverpool Hospital discusses the use of steroid-Campath combinations in p53-deleted cases. In Are We There Yet? you can read an article by Professor Terry Hamblin evaluating the progress made to date on finding a cure for CLL. In another article, Dr. Tim Call, a specialist at Mayo Clinic, Rochester, MN offers some facts and insight into the difficult subject of Familial CLL.
Herbs and Supplements
In our Reference section you will find a new link to a valuable resource from Memorial Sloan-Kettering on herbs, botanicals and other products.
The Virtues of Green Tea
In the section on Phytochemicals, Nutrition and Chemoprevention, you will find an article discussing the anti-cancer activity of green tea polyphenols: Do You Like Drinking Green Tea?
Vitamin D3 or cholecalciferol has multiple functions in human physiology. Some of these functions are critically important in preventing and fighting infections, inflammation and even cancers. Traditionally, the value of this vitamin has been greatly underappreciated — and traditional dosage guidelines are now viewed by experts as seriously inadequate. The risk of hypercalcemia (a serious and even fatal condition) comes into play at high dosage rates: an issue to be carefully considered. There is an impressive body of credible research supporting the new viewpoint. The Vitamin D Council is a well-organized website with comprehensive information on the topic, presented in a straightforward way that patients can understand. The experts behind this organization have impressive credentials. Please do visit this site and read the material presented to decide for yourself whether Vitamin D3 supplementation should be a part of your regimen.
Of course, we have our own collection of articles and Alerts on the subject of Vitamin D3 and its importance in CLL. You can find links to the entire collection on our Vitamin D3 page, or start by reading the article below.
Vitamin D3: Essential for Health
Dosages, Cautions and the Need for Monitoring
Effective Supplementation Can Help Fight Cancer
 Vitamin D is a very potent regulator of cellular biochemistry. We review the research literature on the value of an adequate intake of Vitamin D3 in maintaining general health and in fighting cancer. The literature indicates that the officially recommended dosage (RDA) of the vitamin is barely enough to avoid deficiency diseases and woefully inadequate to provide the multiple health benefits available from this supplement. However, any aggressive supplementation regimen must be carefully monitored and medically supervised since high dosages can lead to systemic toxicity (hypercalcemia) and even death. Our review article, “Vitamin D3: Essential for Health” explores the benefits, raises the caution flags and explains how to benefit from the vitamin without the risk of sun exposure.
Vitamin D is a very potent regulator of cellular biochemistry. We review the research literature on the value of an adequate intake of Vitamin D3 in maintaining general health and in fighting cancer. The literature indicates that the officially recommended dosage (RDA) of the vitamin is barely enough to avoid deficiency diseases and woefully inadequate to provide the multiple health benefits available from this supplement. However, any aggressive supplementation regimen must be carefully monitored and medically supervised since high dosages can lead to systemic toxicity (hypercalcemia) and even death. Our review article, “Vitamin D3: Essential for Health” explores the benefits, raises the caution flags and explains how to benefit from the vitamin without the risk of sun exposure.

The Price of
Military Service
VA Accepts the Agent Orange Connection to CLL
Veterans Please Note –
Court Ordered Retroactive Benefits
Our Agent Orange Update should be of interest to veterans and others inter ested in the scientific evidence of the connection between CLL and Agent Orange and in the formal statements of the VA's approach to this subject.
ested in the scientific evidence of the connection between CLL and Agent Orange and in the formal statements of the VA's approach to this subject.
You can read a recent (April 2025) email on how the VA is treating AO veterans.
OTHER IMPORTANT ARTICLES
Mayo Best Practices
Current Approach to Diagnosis and Management of CLL
What You and Your Oncologist Need to Know about CLL
 We review and comment on an important new paper from the
Mayo Clinic Hematology Team. Authors Shanafelt and Call
provide an extremely useful review of recent changes in
diagnostic methods and risk assessment and their therapeutic
implications for various cytogenetic sub-categories of CLL.
This is a must-read paper for both you and your local oncologist.
Our editorial comment examines areas in which they could have
gone farther in their recommendations. Click here to read
"Need to Know".
We review and comment on an important new paper from the
Mayo Clinic Hematology Team. Authors Shanafelt and Call
provide an extremely useful review of recent changes in
diagnostic methods and risk assessment and their therapeutic
implications for various cytogenetic sub-categories of CLL.
This is a must-read paper for both you and your local oncologist.
Our editorial comment examines areas in which they could have
gone farther in their recommendations. Click here to read
"Need to Know".
What Type of CLL Do You Have?
Analyzing CLL by Risk Categories
Defining Risk "Buckets" as a Framework for Therapy Decisions
 This article, "What Type of CLL Do You Have?",
is a detailed review of a number of important CLL prognostic indicators. It explores a concept for combining the
available indicators to assess progression risk in a given case, grouping patient into buckets
A, B and C. A higher risk category poses trickier problems in picking a treatment strategy but
modern therapeutic options do offer usable approaches.
This article, "What Type of CLL Do You Have?",
is a detailed review of a number of important CLL prognostic indicators. It explores a concept for combining the
available indicators to assess progression risk in a given case, grouping patient into buckets
A, B and C. A higher risk category poses trickier problems in picking a treatment strategy but
modern therapeutic options do offer usable approaches.
FISH-ing for Answers
Chromosomal Aberrations Rule the Game
Getting Your FISH Test Done
 FISH (fluorescence in-situ hybridization) testing can provide key information
to CLL patients and their doctors in making therapy decisions. In "
FISH-ing for Answers" we provide some critical
information that may be useful in getting your very own FISH test done.
FISH (fluorescence in-situ hybridization) testing can provide key information
to CLL patients and their doctors in making therapy decisions. In "
FISH-ing for Answers" we provide some critical
information that may be useful in getting your very own FISH test done.
Familial CLL
Genetic Predisposition and Anticipation
To Know or Not to Know: That Is the Question
 Not the Worst Day of Your Life is a review of the research findings on the
familial aspects of CLL. It is well documented that CLL strikes more than one
person in some families, there is clearly a predisposition to the disease that
has been passed on from one generation to the next in these families.
In addition, the data show that in families where the disease strikes more than one
generation, the children of a CLL patient are likely to be diagnosed with the
disease at an younger age compared to the parent. Finally, we discuss some of
the difficult issues involved in testing children for potential genetic problems.
Not the Worst Day of Your Life is a review of the research findings on the
familial aspects of CLL. It is well documented that CLL strikes more than one
person in some families, there is clearly a predisposition to the disease that
has been passed on from one generation to the next in these families.
In addition, the data show that in families where the disease strikes more than one
generation, the children of a CLL patient are likely to be diagnosed with the
disease at an younger age compared to the parent. Finally, we discuss some of
the difficult issues involved in testing children for potential genetic problems.
Campath - Looking Better and Better
Campath Proves Effective in Early Stage and Difficult Cases
Experts Get Better at Using the Anti-CD 52 Monoclonal
 We review the current status of research and clinical trials
involving Campath-1H. This fully humanized monoclonal antibody
has been approved by the FDA for use in relapsed CLL but current
research reveals that it could have critical application as a
frontline therapy in a subset of high risk cases. It is a powerful
therapeutic agent that is slowly finding wider applicability in
early stage patients. The techniques of administration have
improved and so have the techniques of managing the serious immune
suppression attendant to its use. Our review article,
Campath Looking Better and Better, explores these and other
aspects of Campath therapy.
We review the current status of research and clinical trials
involving Campath-1H. This fully humanized monoclonal antibody
has been approved by the FDA for use in relapsed CLL but current
research reveals that it could have critical application as a
frontline therapy in a subset of high risk cases. It is a powerful
therapeutic agent that is slowly finding wider applicability in
early stage patients. The techniques of administration have
improved and so have the techniques of managing the serious immune
suppression attendant to its use. Our review article,
Campath Looking Better and Better, explores these and other
aspects of Campath therapy.
Prognostic Indicators
Using Modern Tools
Prognosis at Diagnosis - Integrating Molecular Biologic Insights into Clinical Practice
 We examine a critically important new Blood First Edition Paper in
Prognosis at Diagnosis, our review of the article
of the same name from the experts at Mayo Clinic. While much of the research quoted
in their paper has been previously reported, what makes the
paper unique is that it puts it all together in one irrefutable
package: the research, the results, the logic, the recommendations.
I have no doubt this paper will be one of the most influential papers
in CLL, and will be influencing clinical practice for many years to come."
We examine a critically important new Blood First Edition Paper in
Prognosis at Diagnosis, our review of the article
of the same name from the experts at Mayo Clinic. While much of the research quoted
in their paper has been previously reported, what makes the
paper unique is that it puts it all together in one irrefutable
package: the research, the results, the logic, the recommendations.
I have no doubt this paper will be one of the most influential papers
in CLL, and will be influencing clinical practice for many years to come."
Chemotherapy Insight
The "How" Is Often as Important as the "What" in Chemotherapy
Methods of Drug Delivery
 Physiology offers some insights into the effectiveness of various
methods of drug delivery. The same drug can have different effects (and side effects)
when administered using novel techniques. You will find our discussion of this subject in
The "How" Is Often as
Important as the "What".
Physiology offers some insights into the effectiveness of various
methods of drug delivery. The same drug can have different effects (and side effects)
when administered using novel techniques. You will find our discussion of this subject in
The "How" Is Often as
Important as the "What".
Adhesion, Homing and Resistance
Why Peripheral Blood Numbers Do Not Tell the Whole Story — and Why CLL Cells Are Hard to Kill.
It's a Tale of Adhesion Factors, Chemokine Trails, Receptors and Blockers
 Cellular chemistry drives the behaviour of CLL and determines the
characteristics of the different clonal subtypes. We examine a number of critical
aspects such as resistance to therapies, bulkiness of lymph nodes,
infiltration of bone marrow and the support structure for CLL cells
in their preferred environments. We also look at some futuristic
possibilities for therapies in this article,
Adhesion, Homing and Resistance to Therapy.
Cellular chemistry drives the behaviour of CLL and determines the
characteristics of the different clonal subtypes. We examine a number of critical
aspects such as resistance to therapies, bulkiness of lymph nodes,
infiltration of bone marrow and the support structure for CLL cells
in their preferred environments. We also look at some futuristic
possibilities for therapies in this article,
Adhesion, Homing and Resistance to Therapy.
Ctyogenetics
Role of the ATM and TP53 Genes in Disease Progression and Response to Therapy
The Science Behind the Grim Statistics Offers the Hope of Better Therapeutic Strategies
 The TP53 gene and the ATM gene play a critical role in the behavior of CLL cells.
We explore the roles of these genes in the life of a normal cell and their malfunction
in cancer. Modern FISH analysis focusing on these genes helps identify the risk
characteristics and aggressiveness of a given patient's CLL and is therefore an important
prognostic tool. Click here to read
Cytogenetics of ATM and TP53.
The TP53 gene and the ATM gene play a critical role in the behavior of CLL cells.
We explore the roles of these genes in the life of a normal cell and their malfunction
in cancer. Modern FISH analysis focusing on these genes helps identify the risk
characteristics and aggressiveness of a given patient's CLL and is therefore an important
prognostic tool. Click here to read
Cytogenetics of ATM and TP53.
Chronic Pulmonary Inflammation
Major New Study Indicts Role of Pulmonary Inflammation
Deadly Feedback Cycle of Inflammation and CLL
 Widespread anecdotal evidence suggesting pulmonary complications
are a major factor in CLL is now supplanted by solid statistical
data reported in an article in the journal
Cancer by a team led by Dr. Kanti Rai at the Long Island Jewish
Medical Center. They confirm that pneumonia is by far the leading cause
of mortality and morbidity for CLL patients – and the leading
pulmonary complication among patients hospitalized with CLL. Our
article,
Role of Pulmonary Inflammation in CLL, examines the
relationships involved and implications for therapy.
Widespread anecdotal evidence suggesting pulmonary complications
are a major factor in CLL is now supplanted by solid statistical
data reported in an article in the journal
Cancer by a team led by Dr. Kanti Rai at the Long Island Jewish
Medical Center. They confirm that pneumonia is by far the leading cause
of mortality and morbidity for CLL patients – and the leading
pulmonary complication among patients hospitalized with CLL. Our
article,
Role of Pulmonary Inflammation in CLL, examines the
relationships involved and implications for therapy.
Dying to Get a Tan?
The Danger of Squamous Cell Carcinoma in CLL Patients
Things You Can Do to Avoid SCC
 If you have CLL, you have a much higher chance of getting squamous cell carcinoma (SCC), the second most common skin cancer. Further, CLL patients who contract this disease are more likely to have an aggressive form of the disease. We examine some of the possible mechanisms for the onset of this cancer and review a number of steps you can take to protect yourself including a few things you can discuss with your doctor that may help. Click here to read Dying to Get a Tan?
If you have CLL, you have a much higher chance of getting squamous cell carcinoma (SCC), the second most common skin cancer. Further, CLL patients who contract this disease are more likely to have an aggressive form of the disease. We examine some of the possible mechanisms for the onset of this cancer and review a number of steps you can take to protect yourself including a few things you can discuss with your doctor that may help. Click here to read Dying to Get a Tan?
Target Mitochondrion
Promising New Approaches May Level the Playing Field for Bucket C Patients
Attacking Mitochondria in B-CLL Cells
 Several recent research articles have opened up exciting new opportunities
in the treatment of CLL. These feature new small molecule drugs that take
an entirely different approach to targeting CLL cells, bypassing the usual
cellular control points. In this new approach
to treating CLL, it seems to make no difference if you are IgVH mutated or
unmutated, CD38 positive or not, chemo-naïve or have been through the wars
with every chemotherapy drug known to man. In fact, there are some indications
that heavily pretreated and late Rai stage patients may respond better to this
approach. To learn more, read
Exciting New Drugs on the Horizon.
Several recent research articles have opened up exciting new opportunities
in the treatment of CLL. These feature new small molecule drugs that take
an entirely different approach to targeting CLL cells, bypassing the usual
cellular control points. In this new approach
to treating CLL, it seems to make no difference if you are IgVH mutated or
unmutated, CD38 positive or not, chemo-naïve or have been through the wars
with every chemotherapy drug known to man. In fact, there are some indications
that heavily pretreated and late Rai stage patients may respond better to this
approach. To learn more, read
Exciting New Drugs on the Horizon.
AIHA: Auto-Immune Hemolytic Anemia
Important Insight
Causes, Mechanisms & Therapeutic Strategies
 AHIA affects a significant part of the CLL population,
particularly those with advanced disease. Understanding the causes of the
disorder is critical to dealing with it. In
AIHA: Auto-Immune Hemolytic Anemia, we review the relevant research and
make some suggestions.
AHIA affects a significant part of the CLL population,
particularly those with advanced disease. Understanding the causes of the
disorder is critical to dealing with it. In
AIHA: Auto-Immune Hemolytic Anemia, we review the relevant research and
make some suggestions.
Side Effects
EPO Linked to Promotion of Angiogenesis, Tumor Growth and Shorter Survival
The Dark Side of Epoetin
 A very recent study casts light on some negative aspects
of epoetin therapy. In addition to the desired effect of
a rapid increase in red blood cells and hemoglobin and
an improvement in quality of life, these drugs have now
been shown in a carefully designed study to promote
significant angiogenesis and tumor growth. Our
article,
The Dark Side of Epoetin, gives you an overview of
the research on this aspect of an important supporting therapy.
A very recent study casts light on some negative aspects
of epoetin therapy. In addition to the desired effect of
a rapid increase in red blood cells and hemoglobin and
an improvement in quality of life, these drugs have now
been shown in a carefully designed study to promote
significant angiogenesis and tumor growth. Our
article,
The Dark Side of Epoetin, gives you an overview of
the research on this aspect of an important supporting therapy.
Chronic Inflammation
What You Can Do
Interaction between CLL and Chronic Inflammation
 Sinus problems got you down? Here are some conclusions from
recent research in cellular biochemistry. The good news is
that the linkage between inflammation and CLL offers a hint
for a new therapeutic approach. You can read about it in the article
CLL, Chronic Inflammation and What You Can Do About It.
Sinus problems got you down? Here are some conclusions from
recent research in cellular biochemistry. The good news is
that the linkage between inflammation and CLL offers a hint
for a new therapeutic approach. You can read about it in the article
CLL, Chronic Inflammation and What You Can Do About It.
Disease Characteristics
Nature of CLL
Clonal Evolution: How the Disease Grows and Evolves
 In this article we review important new research into the mechanisms by which clonal
B-CLL cells become more aggressive and harder to kill. This is Darwinian selection
at the cellular level. We look at the implications for treatment strategy and offer
a new approach to managing the disease in
Clonal Evolution.
In this article we review important new research into the mechanisms by which clonal
B-CLL cells become more aggressive and harder to kill. This is Darwinian selection
at the cellular level. We look at the implications for treatment strategy and offer
a new approach to managing the disease in
Clonal Evolution.

The
Case of
the Round-Headed
Kid
Notes on a Difficult Case
Things Are Never Easy for the Round-Headed Kid.
Choices are Trickier for Patients with Poor Cytogenetic Aberrations
 Lucy may never have given Charlie Brown that perfect opportunity
to kick a field goal, but we give our own round-headed hero,
Harvey, a hand in figuring out what to do when he gets some bad
news on the cytogenetics front. Faced with a 11q deletion event,
our hero gets down to facing the facts and grappling with some
thorny questions. The saga starts with
The Difficult Case of the Round-headed Kid — and continues
over a number of installments as our hero faces therapy decisions and
disease complications. You will find the entire series of articles in our
RHK Saga.
Lucy may never have given Charlie Brown that perfect opportunity
to kick a field goal, but we give our own round-headed hero,
Harvey, a hand in figuring out what to do when he gets some bad
news on the cytogenetics front. Faced with a 11q deletion event,
our hero gets down to facing the facts and grappling with some
thorny questions. The saga starts with
The Difficult Case of the Round-headed Kid — and continues
over a number of installments as our hero faces therapy decisions and
disease complications. You will find the entire series of articles in our
RHK Saga.

———
Disclaimer: The content of this website is intended for information only and is NOT meant to be medical advice. Please be sure to consult and follow the advice of your doctors on all medical matters.
Copyright Notice:
Copyright © 2025-2007 CLL Topics, Inc. All Rights Reserved.
All materials contained on this site are protected by United States copyright law and may not be reproduced, distributed, transmitted, displayed, published or broadcast without the prior written permission of CLL Topics, Inc. You may not alter or remove any trademark, copyright or other notice from copies of the content.
However, you may download and print material from CLLTopics.org exclusively for your personal, noncommercial use.
———